The Advisory Committee on Immunization Practices (ACIP) annually reviews the recommended Adult Immunization Schedule to ensure that the schedule reflects current recommendations for the licensed vaccines. In October 2009, ACIP approved the Adult Immunization Schedule for 2010, which includes several changes. A bivalent human papillomavirus vaccine (HPV2) was licensed for use in females in October 2009. ACIP recommends vaccination of females with either HPV2 or the quadrivalent human papillomavirus vaccine (HPV4). HPV4 was licensed for use in males in October 2009, and ACIP issued a permissive recommendation for use in males. Introductory sentences were added to the footnotes for measles, mumps, rubella, influenza, pneumococcal, hepatitis A, hepatitis B, and meningococcal vaccines. Clarifications were made to the footnotes for measles, mumps, rubella, influenza, hepatitis A, meningococcal, and Haemophilus influenza type b vaccines, and schedule information was added to the hepatitis B vaccine footnote.
The Recommended Adult Immunization Schedule has been approved by the Advisory Committee on Immunization Practices, the American Academy of Family Physicians, the American College of Obstetricians and Gynecologists, and the American College of Physicians.
Suggested citation: Centers for Disease Control and Prevention. Recommended adult immunization schedule---United States, 2010. MMWR 2010;59(1).
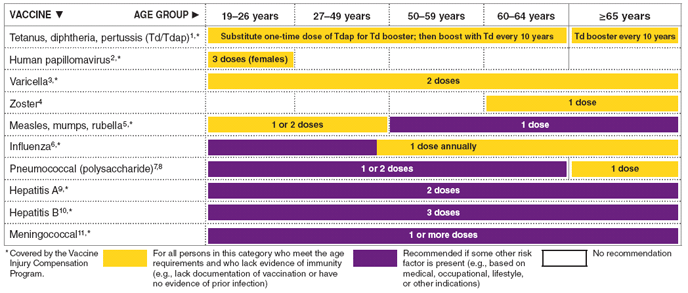
FIGURE 1. Recommended adult immunization schedule, by vaccine and age group - United States, 2010
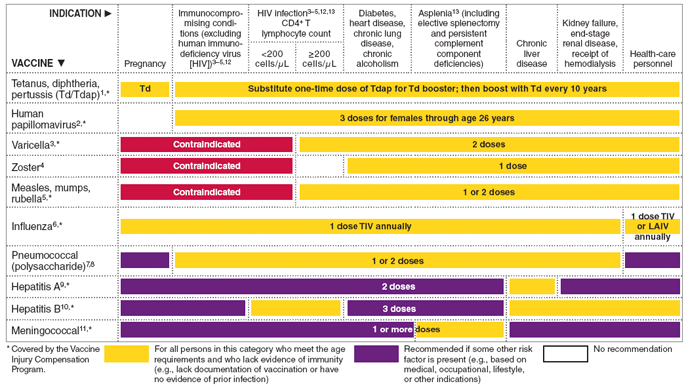
FIGURE 2. Vaccines that might be indicated for adults, based on medical and other indications - United States, 2010
- Tetanus, diphtheria, and acellular pertussis (Td/Tdap) vaccination
Tdap should replace a single dose of Td for adults aged 19--64 years who have not received a dose of Tdap previously.
Adults with uncertain or incomplete history of primary vaccination series with tetanus and diphtheria toxoid-containing vaccines should begin or complete a primary vaccination series. A primary series for adults is 3 doses of tetanus and diphtheria toxoid-containing vaccines; administer the first 2 doses at least 4 weeks apart and the third dose 6--12 months after the second; Tdap can substitute for any one of the doses of Td in the 3-dose primary series. The booster dose of tetanus and diphtheria toxoid-containing vaccine should be administered to adults who have completed a primary series and if the last vaccination was received ≥10 years previously. Tdap or Td vaccine may be used, as indicated.
If a woman is pregnant and received the last Td vaccination ≥10 years previously, administer Td during the second or third trimester. If the woman received the last Td vaccination <10 years previously, administer Tdap during the immediate postpartum period. A dose of Tdap is recommended for postpartum women, close contacts of infants aged <12 months, and all health-care personnel with direct patient contact if they have not previously received Tdap. An interval as short as 2 years from the last Td vaccination is suggested; shorter intervals can be used. Td may be deferred during pregnancy and Tdap substituted in the immediate postpartum period, or Tdap can be administered instead of Td to a pregnant woman.
Consult the ACIP statement for recommendations for giving Td as prophylaxis in wound management.
- Human papillomavirus (HPV) vaccination
HPV vaccination is recommended at age 11 or 12 years with catch-up vaccination at ages 13 through 26 years.
Ideally, vaccine should be administered before potential exposure to HPV through sexual activity; however, females who are sexually active should still be vaccinated consistent with age-based recommendations. Sexually active females who have not been infected with any of the four HPV vaccine types (types 6, 11, 16, 18, all of which HPV4 prevents) or any of the two HPV vaccine types (types 16 and 18, both of which HPV2 prevents) receive the full benefit of the vaccination. Vaccination is less beneficial for females who have already been infected with one or more of the HPV vaccine types. HPV4 or HPV2 can be administered to persons with a history of genital warts, abnormal Papanicolaou test, or positive HPV DNA test, because these conditions are not evidence of prior infection with all vaccine HPV types.
HPV4 may be administered to males aged 9 through 26 years to reduce their likelihood of acquiring genital warts. HPV4 would be most effective when administered before exposure to HPV through sexual contact.
A complete series for either HPV4 or HPV2 consists of 3 doses. The second dose should be administered 1--2 months after the first dose; the third dose should be administered 6 months after the first dose.
Although HPV vaccination is not specifically recommended for persons with the medical indications described in Figure 2, "Vaccines that might be indicated for adults based on medical and other indications," it may be administered to these persons because the HPV vaccine is not a live-virus vaccine. However, the immune response and vaccine efficacy might be less for persons with the medical indications described in Figure 2 than in persons who do not have the medical indications described or who are immunocompetent. Health-care personnel are not at increased risk because of occupational exposure and should be vaccinated consistent with age-based recommendations.
- Varicella vaccination
All adults without evidence of immunity to varicella should receive 2 doses of single-antigen varicella vaccine if not previously vaccinated or the second dose if they have received only 1 dose, unless they have a medical contraindication. Special consideration should be given to those who 1) have close contact with persons at high risk for severe disease (e.g., health-care personnel and family contacts of persons with immunocompromising conditions) or 2) are at high risk for exposure or transmission (e.g., teachers; child-care employees; residents and staff members of institutional settings, including correctional institutions; college students; military personnel; adolescents and adults living in households with children; nonpregnant women of childbearing age; and international travelers).
Evidence of immunity to varicella in adults includes any of the following: 1) documentation of 2 doses of varicella vaccine at least 4 weeks apart; 2) U.S.-born before 1980 (although for health-care personnel and pregnant women, birth before 1980 should not be considered evidence of immunity); 3) history of varicella based on diagnosis or verification of varicella by a health-care provider (for a patient reporting a history of or having an atypical case, a mild case, or both, health-care providers should seek either an epidemiologic link with a typical varicella case or to a laboratory-confirmed case or evidence of laboratory confirmation, if it was performed at the time of acute disease); 4) history of herpes zoster based on diagnosis or verification of herpes zoster by a health-care provider; or 5) laboratory evidence of immunity or laboratory confirmation of disease.
Pregnant women should be assessed for evidence of varicella immunity. Women who do not have evidence of immunity should receive the first dose of varicella vaccine upon completion or termination of pregnancy and before discharge from the health-care facility. The second dose should be administered 4--8 weeks after the first dose.
- Herpes zoster vaccination
A single dose of zoster vaccine is recommended for adults aged ≥60 years regardless of whether they report a prior episode of herpes zoster. Persons with chronic medical conditions may be vaccinated unless their condition constitutes a contraindication.
- Measles, mumps, rubella (MMR) vaccination
Adults born before 1957 generally are considered immune to measles and mumps.
Measles component: Adults born during or after 1957 should receive 1 or more doses of MMR vaccine unless they have 1) a medical contraindication; 2) documentation of vaccination with 1 or more doses of MMR vaccine; 3) laboratory evidence of immunity; or 4) documentation of physician-diagnosed measles.
A second dose of MMR vaccine, administered 4 weeks after the first dose, is recommended for adults who 1) have been recently exposed to measles or are in an outbreak setting; 2) have been vaccinated previously with killed measles vaccine; 3) have been vaccinated with an unknown type of measles vaccine during 1963--1967; 4) are students in postsecondary educational institutions; 5) work in a health-care facility; or 6) plan to travel internationally.
Mumps component: Adults born during or after 1957 should receive 1 dose of MMR vaccine unless they have 1) a medical contraindication; 2) documentation of vaccination with 1 or more doses of MMR vaccine; 3) laboratory evidence of immunity; or 4) documentation of physician-diagnosed mumps.
A second dose of MMR vaccine, administered 4 weeks after the first dose, is recommended for adults who 1) live in a community experiencing a mumps outbreak and are in an affected age group; 2) are students in postsecondary educational institutions; 3) work in a health-care facility; or 4) plan to travel internationally.
Rubella component: 1 dose of MMR vaccine is recommended for women who do not have documentation of rubella vaccination, or who lack laboratory evidence of immunity. For women of childbearing age, regardless of birth year, rubella immunity should be determined, and women should be counseled regarding congenital rubella syndrome. Women who do not have evidence of immunity should receive MMR vaccine upon completion or termination of pregnancy and before discharge from the health-care facility.
Health-care personnel born before 1957: For unvaccinated health-care personnel born before 1957 who lack laboratory evidence of measles, mumps, and/or rubella immunity or laboratory confirmation of disease, health-care facilities should consider vaccinating personnel with 2 doses of MMR vaccine at the appropriate interval (for measles and mumps) and 1 dose of MMR vaccine (for rubella), respectively.
During outbreaks, health-care facilities should recommend that unvaccinated health-care personnel born before 1957, who lack laboratory evidence of measles, mumps, and/or rubella immunity or laboratory confirmation of disease, receive 2 doses of MMR vaccine during an outbreak of measles or mumps, and 1 dose during an outbreak of rubella.
Complete information about evidence of immunity is available at http://www.cdc.gov/vaccines/recs/provisional/default.htm.
- Seasonal influenza vaccination
Vaccinate all persons aged ≥50 years and any younger persons who would like to decrease their risk for influenza. Vaccinate persons aged 19 through 49 years with any of the following indications.
Medical: Chronic disorders of the cardiovascular or pulmonary systems, including asthma; chronic metabolic diseases (including diabetes mellitus); renal or hepatic dysfunction, hemoglobinopathies, or immunocompromising conditions (including immunocompromising conditions caused by medications or HIV); cognitive, neurologic, or neuromuscular disorders; and pregnancy during the influenza season. No data exist on the risk for severe or complicated influenza disease among persons with asplenia; however, influenza is a risk factor for secondary bacterial infections that can cause severe disease among persons with asplenia.
Occupational: All health-care personnel, including those employed by long-term care and assisted-living facilities, and caregivers of children aged <5 years.
Other: Residents of nursing homes and other long-term care and assisted-living facilities; persons likely to transmit influenza to persons at high risk (e.g., in-home household contacts and caregivers of children aged <5 years, persons aged ≥50 years, and persons of all ages with high-risk conditions).
Healthy, nonpregnant adults aged <50 years without high-risk medical conditions who are not contacts of severely immunocompromised persons in special-care units may receive either intranasally administered live, attenuated influenza vaccine (FluMist) or inactivated vaccine. Other persons should receive the inactivated vaccine.
- Pneumococcal polysaccharide (PPSV) vaccination
Vaccinate all persons with the following indications.
Medical: Chronic lung disease (including asthma); chronic cardiovascular diseases; diabetes mellitus; chronic liver diseases, cirrhosis; chronic alcoholism; functional or anatomic asplenia (e.g., sickle cell disease or splenectomy [if elective spletnectomy is planned, vaccinate at least 2 weeks before surgery]); immunocompromising conditions (including chronic renal failure or nephrotic syndrome); and cochlear implants and cerebrospinal fluid leaks. Vaccinate as close to HIV diagnosis as possible.
Other: Residents of nursing homes or long-term care facilities and persons who smoke cigarettes. Routine use of PPSV is not recommended for American Indians/Alaska Natives or persons aged <65 years unless they have underlying medical conditions that are PPSV indications. However, public health authorities may consider recommending PPSV for American Indians/Alaska Natives and persons aged 50 through 64 years who are living in areas where the risk for invasive pneumococcal disease is increased.
- Revaccination with PPSV
One-time revaccination after 5 years is recommended for persons with chronic renal failure or nephrotic syndrome; functional or anatomic asplenia (e.g., sickle cell disease or splenectomy); and for persons with immunocompromising conditions. For persons aged ≥65 years, one-time revaccination is recommended if they were vaccinated ≥5 years previously and were aged <65 years at the time of primary vaccination.
- Hepatitis A vaccination
Vaccinate persons with any of the following indications and any person seeking protection from hepatitis A virus (HAV) infection.
Behavioral: Men who have sex with men and persons who use injection drugs.
Occupational: Persons working with HAV-infected primates or with HAV in a research laboratory setting.
Medical: Persons with chronic liver disease and persons who receive clotting factor concentrates.
Other: Persons traveling to or working in countries that have high or intermediate endemicity of hepatitis A (a list of countries is available at http://wwwn.cdc.gov/travel/contentdiseases.aspx).
Unvaccinated persons who anticipate close personal contact (e.g., household contact or regular babysitting) with an international adoptee from a country of high or intermediate endemicity during the first 60 days after arrival of the adoptee in the United States should consider vaccination. The first dose of the 2-dose hepatitis A vaccine series should be administered as soon as adoption is planned, ideally ≥2 weeks before the arrival of the adoptee.
Single-antigen vaccine formulations should be administered in a 2-dose schedule at either 0 and 6--12 months (Havrix), or 0 and 6--18 months (Vaqta). If the combined hepatitis A and hepatitis B vaccine (Twinrix) is used, administer 3 doses at 0, 1, and 6 months; alternatively, a 4-dose schedule, administered on days 0, 7, and 21--30 followed by a booster dose at month 12 may be used.
- Hepatitis B vaccination
Vaccinate persons with any of the following indications and any person seeking protection from hepatitis B virus (HBV) infection.
Behavioral: Sexually active persons who are not in a long-term, mutually monogamous relationship (e.g., persons with more than one sex partner during the previous 6 months); persons seeking evaluation or treatment for a sexually transmitted disease (STD); current or recent injection-drug users; and men who have sex with men.
Occupational: Health-care personnel and public-safety workers who are exposed to blood or other potentially infectious body fluids.
Medical: Persons with end-stage renal disease, including patients receiving hemodialysis; persons with HIV infection; and persons with chronic liver disease.
Other: Household contacts and sex partners of persons with chronic HBV infection; clients and staff members of institutions for persons with developmental disabilities; and international travelers to countries with high or intermediate prevalence of chronic HBV infection (a list of countries is available at http://wwwn.cdc.gov/travel/contentdiseases.aspx).
Hepatitis B vaccination is recommended for all adults in the following settings: STD treatment facilities; HIV testing and treatment facilities; facilities providing drug-abuse treatment and prevention services; health-care settings targeting services to injection-drug users or men who have sex with men; correctional facilities; end-stage renal disease programs and facilities for chronic hemodialysis patients; and institutions and nonresidential day-care facilities for persons with developmental disabilities.
Administer or complete a 3-dose series of hepatitis B vaccine to those persons not previously vaccinated. The second dose should be administered 1 month after the first dose; the third dose should be administered at least 2 months after the second dose (and at least 4 months after the first dose). If the combined hepatitis A and hepatitis B vaccine (Twinrix) is used, administer 3 doses at 0, 1, and 6 months; alternatively, a 4-dose schedule, administered on days 0, 7, and 21--30 followed by a booster dose at month 12 may be used.
Adult patients receiving hemodialysis or with other immunocompromising conditions should receive 1 dose of 40 µg/mL (Recombivax HB) administered on a 3-dose schedule or 2 doses of 20 µg/mL (Engerix-B) administered simultaneously on a 4-dose schedule at 0, 1, 2, and 6 months.
- Meningococcal vaccination
Meningococcal vaccine should be administered to persons with the following indications.
Medical: Adults with anatomic or functional asplenia, or persistent complement component deficiencies.
Other: First-year college students living in dormitories; microbiologists routinely exposed to isolates of Neisseria meningitidis; military recruits; and persons who travel to or live in countries in which meningococcal disease is hyperendemic or epidemic (e.g., the "meningitis belt" of sub-Saharan Africa during the dry season [December through June]), particularly if their contact with local populations will be prolonged. Vaccination is required by the government of Saudi Arabia for all travelers to Mecca during the annual Hajj.
Meningococcal conjugate vaccine (MCV4) is preferred for adults with any of the preceding indications who are aged ≤55 years; meningococcal polysaccharide vaccine (MPSV4) is preferred for adults aged ≥56 years. Revaccination with MCV4 after 5 years is recommended for adults previously vaccinated with MCV4 or MPSV4 who remain at increased risk for infection (e.g., adults with anatomic or functional asplenia). Persons whose only risk factor is living in on-campus housing are not recommended to receive an additional dose.
- Immunocompromising conditions
Inactivated vaccines generally are acceptable (e.g., pneumococcal, meningococcal, influenza [inactivated influenza vaccine]) and live vaccines generally are avoided in persons with immune deficiencies or immunocompromising conditions. Information on specific conditions is available at http://www.cdc.gov/vaccines/pubs/acip-list.htm.
- Selected conditions for which Haemophilus influenzae type b (Hib) vaccine may be used
Hib vaccine generally is not recommended for persons aged ≥5 years. No efficacy data are available on which to base a recommendation concerning use of Hib vaccine for older children and adults. However, studies suggest good immunogenicity in patients who have sickle cell disease, leukemia, or HIV infection or who have had a splenectomy. Administering 1 dose of Hib vaccine to these high-risk persons who have not previously received Hib vaccine is not contraindicated.
 ShareCompartir
ShareCompartir