Intervals Between PCV13 and PPSV23 Vaccines: Recommendations of the Advisory Committee on Immunization Practices (ACIP)
Please note: An erratum has been published for this article. To view the erratum, please click here.
, MD1,2; , MD3,4; , MPH1; , MSPH1; , MD1; , MD1; , MPH1
Two pneumococcal vaccines are currently licensed for use in the United States: the 13-valent pneumococcal conjugate vaccine (PCV13 [Prevnar 13, Wyeth Pharmaceuticals, Inc., a subsidiary of Pfizer Inc.]) and the 23-valent pneumococcal polysaccharide vaccine (PPSV23 [Pneumovax 23, Merck and Co., Inc.]). The Advisory Committee on Immunization Practices (ACIP) currently recommends that a dose of PCV13 be followed by a dose of PPSV23 in all adults aged ≥65 years who have not previously received pneumococcal vaccine and in persons aged ≥2 years who are at high risk for pneumococcal disease because of underlying medical conditions (Table) (1–4). The recommended intervals between PCV13 and PPSV23 given in series differ by age and risk group and the order in which the two vaccines are given (1–4).
On June 25, 2015, ACIP changed the recommended interval between PCV13 followed by PPSV23 (PCV13–PPSV23 sequence) from 6–12 months to ≥1 year for immunocompetent adults aged ≥65 years. Recommended intervals for all other age and risk groups remain unchanged. This report outlines the rationale for this change and summarizes the evidence considered by ACIP to make this recommendation.
In August 2014, ACIP recommended routine use of a dose of PCV13 followed by a dose of PPSV23 6–12 months later among immunocompetent adults aged ≥65 years (1). Adults aged ≥65 years with immunocompromising conditions, functional or anatomic asplenia, cerebrospinal fluid (CSF) leaks, or cochlear implants are recommended to receive PCV13 first, followed by PPSV23 ≥8 weeks later (2). ACIP also recommended that all adults aged ≥65 years who already received PPSV23 should receive a dose of PCV13 ≥1 year after receipt of PPSV23 (PPSV23–PCV13 sequence). The difference in the recommended interval depending on the order in which the two vaccines were given added significant complexity to the recommendation and created implementation challenges for this age group. To simplify the recommendations, ACIP reviewed existing data to evaluate potential areas for harmonization of recommended dosing intervals. Specifically, ACIP assessed whether available evidence would support changing the recommended interval for the PCV13–PPSV23 sequence for immunocompetent adults aged ≥65 years from 6–12 months to ≥1 year and thus be harmonized with the recommended interval for the PPSV23–PCV13 sequence in the same age group.
No clinical studies evaluating efficacy of the two vaccines given in series are available. Therefore, current recommendations are based on best available evidence from immunogenicity studies. The Grading of Recommendations, Assessment, Development, and Evaluation (GRADE) framework was used by ACIP to formulate the existing recommendations for immunocompromised children (http://www.cdc.gov/vaccines/acip/recs/grade/pneumo-immuno-child.html), immunocompromised adults (http://www.cdc.gov/vaccines/acip/recs/grade/pneumo-immuno-adults.html), and adults ≥65 years (http://www.cdc.gov/vaccines/acip/recs/grade/pneumo-vac-adult.html) (1–3). No new evidence was available to inform harmonization of intervals; therefore, the GRADE process was not repeated. In addition, the immunogenicity studies were not designed to evaluate the optimal interval between the two vaccines. When both PCV13 and PPSV23 are to be administered, PCV13 is recommended before PPSV23, based on studies demonstrating a better response to serotypes common to both vaccines when PCV was given first (5–7).
Studies evaluating the immune response to a conjugate vaccine (PCV7 or PCV13) followed by the polysaccharide vaccine (PCV–PPSV23 sequence) at intervals of 2, 6, or 12 months or 3–4 years demonstrated that following the PPSV23 dose, antibody levels against serotypes common to both vaccines were higher than the pre-PCV baseline (5,6,8–13). Eight studies compared immune responses among immunocompetent adults aged ≥50 years after a PCV–PPSV23 sequence with responses following PCV or PPSV23 administered alone (5,6,8–13). Four studies showed that antibody responses (measured by opsonophagocytic activity [OPA] or immunoglobulin G [IgG] levels or both) following PCV7–PPSV23 doses given 6 months apart were better than or equivalent to responses following PCV7 or PPSV23 alone for most pneumococcal serotypes measured (6,8,11,12). Another study showed that a 1-year interval between receipt of conjugate vaccine and polysaccharide vaccine (PCV7–PPSV23 sequence) also led to improved immune responses compared with those following a single PPSV23 dose (13). Comparison of antibody responses after a PCV13–PPSV23 sequence to responses following PCV13 or PPSV23 alone, across two studies with intervals of 1 year and 3–4 years between the two vaccines, indicated that the responses to a larger number of serotypes are improved with a 3–4 year interval compared with a 1-year interval (5,9). One study among pneumococcal vaccine–naïve Alaska Native adults aged 55–70 years included direct comparison between intervals of 2 months and 6 months between receipt of PCV7 and PPSV23. No differences in the immune responses were observed; however, the group with a 2-month interval between doses reported more injection site swelling than the group with a 6-month interval (10).
In summary, these studies of PCV–PPSV23 sequence among immunocompetent adults suggest that 1) shorter intervals (e.g., 8 weeks), may be associated with increased local reactogenicity when compared with longer intervals, and 2) longer intervals (e.g., ≥1 year) may lead to an improved immune response against serotypes in both vaccines compared with a single dose of PCV13 or PPSV23. Additionally, changing the recommended interval for the PCV13–PPSV23 sequence to ≥1 year would allow the recommended interval for immunocompetent adults aged ≥65 years to be the same, regardless of the order in which the two vaccines are given (Box).
ACIP considered additional factors when determining whether a change to the intervals is warranted. These factors include the risk window for protection against disease caused by serotypes unique to PPSV23, the timing for the next visit to the vaccination provider, as well as revised Centers for Medicare and Medicaid Services (CMS) regulations allowing for coverage of the two pneumococcal vaccines when given in series and administered 1 year apart. Approximately 40% of invasive pneumococcal disease among adults aged ≥65 years is caused by serotypes unique to PPSV23. The potential change in the interval for the PCV13–PPSV23 sequence is most likely to affect the youngest adults in this age group who are more likely to be pneumococcal vaccine naïve. The incidence of disease caused by serotypes unique to PPSV23 is lowest among these adults (14).
The 2012 National Health Interview Survey results suggest that >85% of adults in the United States aged ≥65 years had at least one encounter with a health professional within the preceding 6 months, and >93% within the preceding year (15).Therefore, the 1-year interval would offer the best opportunity for the majority of eligible adults aged ≥65 years to receive the recommended pneumococcal vaccine series during their existing healthcare encounters and not require an extra visit.
The recently revised CMS regulations for pneumococcal vaccines allow for Medicare coverage of a different, second pneumococcal vaccine 1 year after the first vaccine is given (16). The change in the ACIP recommended interval for the PCV13–PPSV23 sequence would make ACIP recommendations consistent with the current Medicare policy.
Recommended intervals between PCV13 and PPSV23 for persons aged ≥2 years with medical indications to receive both vaccines remain unchanged (Table). PPSV23 is recommended to be given ≥8 weeks after PCV13 for children and adults aged ≥19 years with certain underlying medical conditions (including adults aged ≥65 years with immunocompromising conditions, functional or anatomic asplenia, CSF leaks, or cochlear implants). Studies among HIV-positive adults evaluating the immune response to PPSV23 administered 4 or 8 weeks after PCV7 showed statistically significant increases in antibody levels compared with response to PPSV23 alone (17,18). The currently recommended 8-week interval minimizes the risk window for invasive pneumococcal disease caused by serotypes unique to PPSV23 in these highly vulnerable groups.
ACIP Recommendations for Intervals Between PCV13 Followed by PPSV23 for Immunocompetent Adults Aged ≥65 Years
For immunocompetent adults aged ≥65 years who have not previously received pneumococcal vaccine, ACIP makes the following recommendation for intervals between PCV13 followed by PPSV23: A dose of PPSV23 should be given ≥1 year following a dose of PCV13. The two vaccines should not be co-administered. If a dose of PPSV23 is inadvertently given earlier than the recommended interval, the dose need not be repeated.
Adverse events occurring after administration of any vaccine should be reported to the Vaccine Adverse Events Reporting System (VAERS). Reports can be submitted to VAERS online, by facsimile, or by mail. More information about VAERS is available by calling 1–800–822–7967 (toll-free) or online at http://vaers.hhs.gov.
Acknowledgments
ACIP members (membership roster for July 2014–June 2015 available at: http://www.cdc.gov/vaccines/acip/committee/members-archive.html); ACIP Pneumococcal Work Group.
1National Center for Immunization and Respiratory Diseases, CDC; 2Epidemic Intelligence Service, CDC; 3Pneumococcal Vaccines Working Group Chair, Advisory Committee on Immunization Practices; 4Department of Medicine, University of Rochester School of Medicine and Dentistry, Rochester, New York.
Corresponding author: Tamara Pilishvili, tpilishvili@cdc.gov, 404-639-2215.
References
- Tomczyk S, Bennett NM, Stoecker C, et al. Use of 13-valent pneumococcal conjugate vaccine and 23-valent pneumococcal polysaccharide vaccine among adults aged ≥65 years: recommendations of the Advisory Committee on Immunization Practices (ACIP). MMWR Morb Mortal Wkly Rep 2014;63:822–5.
- CDC. Use of 13-valent pneumococcal conjugate vaccine and 23-valent pneumococcal polysaccharide vaccine for adults with immunocompromising conditions: recommendations of the Advisory Committee on Immunization Practices (ACIP). MMWR Morb Mortal Wkly Rep 2012;61:816–9.
- CDC. Use of 13-valent pneumococcal conjugate vaccine and 23-valent pneumococcal polysaccharide vaccine among children aged 6–18 years with immunocompromising conditions: recommendations of the Advisory Committee on Immunization Practices (ACIP). MMWR Morb Mortal Wkly Rep 2013;62:521–4.
- Nuorti JP, Whitney CG. Prevention of pneumococcal disease among infants and children—use of 13-valent pneumococcal conjugate vaccine and 23-valent pneumococcal polysaccharide vaccine— recommendations of the Advisory Committee on Immunization Practices (ACIP). MMWR Recomm Rep 2010;59(No. RR-11):1–18.
- Greenberg RN, Gurtman A, Frenck RW, et al. Sequential administration of 13-valent pneumococcal conjugate vaccine and 23-valent pneumococcal polysaccharide vaccine in pneumococcal vaccine-naïve adults 60–64 years of age. Vaccine 2014;32:2364–74.
- Musher DM, Rueda AM, Nahm MH, Graviss EA, Rodriguez-Barradas MC. Initial and subsequent response to pneumococcal polysaccharide and protein-conjugate vaccines administered sequentially to adults who have recovered from pneumococcal pneumonia. J Infect Dis 2008;198:1019–27.
- Blum MD, Dagan R, Mendelman PM, et al. A comparison of multiple regimens of pneumococcal polysaccharide-meningococcal outer membrane protein complex conjugate vaccine and pneumococcal polysaccharide vaccine in toddlers. Vaccine 2000;18:2359–67.
- Goldblatt D, Southern J, Andrews N, et al. The immunogenicity of 7-valent pneumococcal conjugate vaccine versus 23-valent polysaccharide vaccine in adults aged 50–80 years. Clin Infect Dis 2009;49:1318–25.
- Jackson LA, Gurtman A, van Cleeff M, et al. Influence of initial vaccination with 13-valent pneumococcal conjugate vaccine or 23-valent pneumococcal polysaccharide vaccine on anti-pneumococcal responses following subsequent pneumococcal vaccination in adults 50 years and older. Vaccine 2013;31:3594–602.
- Miernyk KM, Butler JC, Bulkow LR, et al. Immunogenicity and reactogenicity of pneumococcal polysaccharide and conjugate vaccines in Alaska native adults 55–70 years of age. Clin Infect Dis 2009;49:241–8.
- Lazarus R, Clutterbuck E, Yu LM, et al. A randomized study comparing combined pneumococcal conjugate and polysaccharide vaccination schedules in adults. Clin Infect Dis 2011;52:736–42.
- Macintyre CR, Ridda I, Gao Z, et al. A randomized clinical trial of the immunogenicity of 7-valent pneumococcal conjugate vaccine compared to 23-valent polysaccharide vaccine in frail, hospitalized elderly. PLoS One 2014;9:e94578.
- de Roux A, Schmöle-Thoma B, Siber GR, et al. Comparison of pneumococcal conjugate polysaccharide and free polysaccharide vaccines in elderly adults: conjugate vaccine elicits improved antibacterial immune responses and immunological memory. Clin Infect Dis 2008;46:1015–23.
- CDC. Active bacterial core surveilance (ABCs). Atlanta, GA: US Department of Health and Human Services, CDC; 2013. Available from: http://www.cdc.gov/abcs/reports-findings/survreports/spneu13.html.
- Blackwell DL, Lucas JW, Clarke TC. Summary health statistics for US adults: national health interview survey, 2012. Vital Health Stat 10 2014;10:1–161.
- Centers for Medicare and Medicaid Services. Modifications to Medicare part B coverage of pneumococcal vaccinations. Washington, DC: Department of Health and Human Services, Centers for Medicare and Medicaid Services; 2014. Available at https://www.cms.gov/Outreach-and-Education/Medicare-Learning-Network-MLN/MLNMattersArticles/Downloads/MM9051.pdf.
- Feikin DR, Elie CM, Goetz MB, et al. Randomized trial of the quantitative and functional antibody responses to a 7-valent pneumococcal conjugate vaccine and/or 23-valent polysaccharide vaccine among HIV-infected adults. Vaccine 2001;20:545–53.
- Lesprit P, Pédrono G, Molina JM, et al. Immunological efficacy of a prime-boost pneumococcal vaccination in HIV-infected adults. AIDS 2007;21:2425–34.
Summary
What is currently recommended?
The Advisory Committee on Immunization Practices (ACIP) currently recommends that both 13-valent pneumococcal conjugate vaccine (PCV13) and 23-valent pneumococcal polysaccharide vaccine (PPSV23) be given to all immunocompetent adults aged ≥65 years. ACIP recommends that PCV13 be given first followed by PPSV23 6–12 months later. ACIP also recommends that adults aged ≥65 years who already received a dose of PPSV23, should also receive a dose of PCV13 ≥1 year after the dose of PPSV23. Among persons aged ≥2 years with medical indications to receive both PCV13 and PPSV23 in a series, including adults aged ≥65 years with immunocompromising conditions, functional or anatomic asplenia, cochlear implants, or cerebrospinal fluid leaks, a dose of PPSV23 should be given ≥8 weeks after a dose of PCV13.
Why are the recommendations being modified now?
To simplify the recommendations for PCV13 and PPSV23 use among immunocompetent adults aged ≥65 years, ACIP recommended harmonization of recommended intervals between PCV13 and PPSV23 regardless of the order in which the two vaccines are given.
What are the new recommendations?
ACIP recommends that both PCV13 and PPSV23 be given in series to adults aged ≥65 years. A dose of PCV13 should be given first followed by a dose of PPSV23 at least 1 year later to immunocompetent adults aged ≥65 years. The two vaccines should not be co-administered. If a dose of PPSV23 is inadvertently given earlier than the recommended interval, the dose need not be repeated.
Recommendations for routine use of vaccines in children, adolescents and adults are developed by the Advisory Committee on Immunization Practices (ACIP). ACIP is chartered as a federal advisory committee to provide expert external advice and guidance to the Director of the Centers for Disease Control and Prevention (CDC) on use of vaccines and related agents for the control of vaccine-preventable diseases in the civilian population of the United States. Recommendations for routine use of vaccines in children and adolescents are harmonized to the greatest extent possible with recommendations made by the American Academy of Pediatrics (AAP), the American Academy of Family Physicians (AAFP), and the American College of Obstetricians and Gynecologists (ACOG). Recommendations for routine use of vaccines in adults are harmonized with recommendations of AAFP, ACOG, and the American College of Physicians (ACP). ACIP recommendations approved by the CDC Director become agency guidelines on the date published in the Morbidity and Mortality Weekly Report (MMWR). Additional information about ACIP is available at http://www.cdc.gov/vaccines/acip.
BOX. Recommended intervals for sequential use of PCV13 and PPSV23 for immunocompetent adults aged ≥65 years — Advisory Committee on Immunization Practices, United States
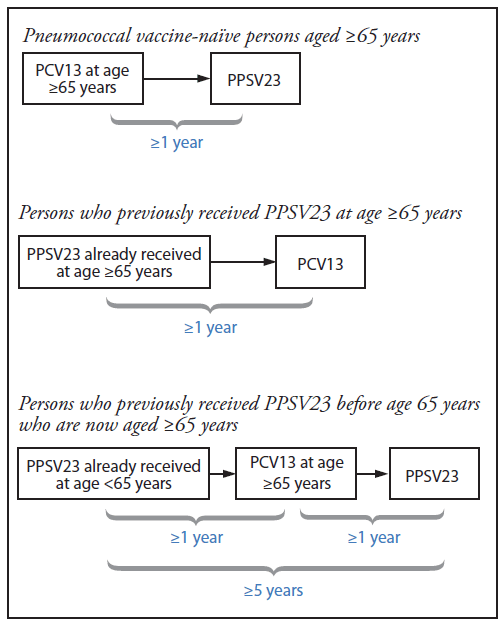
Abbreviations: PCV13 = 13-valent pneumococcal conjugate vaccine; PPSV23 = 23-valent pneumococcal polysaccharide vaccine.
Notes: For adults aged ≥65 years with immunocompromising conditions, functional or anatomic asplenia, cerebrospinal fluid leaks, or cochlear implants, the recommended interval between PCV13 followed by PPSV23 is ≥8 weeks. For those for who previously received PPSV23 when aged <65 years and for whom an additional dose of PPSV23 is indicated when aged ≥65 years, this subsequent PPSV23 dose should be given ≥1 year after PCV13 and ≥5 years after the most recent dose of PPSV23.
Alternate Text: The above box outlines the Advisory Committee on Immunization Practices recommended intervals for sequential use of 13-valent pneumococcal conjugate vaccine and 23-valent pneumococcal polysaccharide vaccine for immunocompetent adults aged ≥65 years in the United States.
Use of trade names and commercial sources is for identification only and does not imply endorsement by the U.S. Department of
Health and Human Services.
References to non-CDC sites on the Internet are
provided as a service to MMWR readers and do not constitute or imply
endorsement of these organizations or their programs by CDC or the U.S.
Department of Health and Human Services. CDC is not responsible for the content
of pages found at these sites. URL addresses listed in MMWR were current as of
the date of publication.
All MMWR HTML versions of articles are electronic conversions from typeset documents.
This conversion might result in character translation or format errors in the HTML version.
Users are referred to the electronic PDF version (http://www.cdc.gov/mmwr)
and/or the original MMWR paper copy for printable versions of official text, figures, and tables.
An original paper copy of this issue can be obtained from the Superintendent of Documents, U.S.
Government Printing Office (GPO), Washington, DC 20402-9371;
telephone: (202) 512-1800. Contact GPO for current prices.
**Questions or messages regarding errors in formatting should be addressed to
mmwrq@cdc.gov.
 ShareCompartir
ShareCompartir


