Notes from the Field: Pneumonia Associated with an Influenza A H3 Outbreak at a Skilled Nursing Facility — Florida, 2014
, MD1,2; , MPH2; , MPH3; , MPH2; , MPH2; , PhD2; , PhD2; , MPH4; , MD2
In December 2014, the Florida Department of Health, Bureau of Epidemiology, was notified that 18 of 95 (19%) residents at a skilled nursing facility had radiographic evidence of pneumonia and were being treated with antibiotics. Two residents were hospitalized, one of whom died. A second resident died at the facility. The Florida Department of Health conducted an outbreak investigation to ascertain all cases through active case finding, identify the etiology, provide infection control guidance, and recommend treatment or prophylaxis, if indicated.
An outbreak-associated case was defined as the onset of fever or respiratory illness in a nursing facility resident or staff member from November 29–December 29, 2014. Overall, 50 persons, including 44 (46%) residents and six (8%) of 75 staff members met the case definition. The earliest reported onset date was November 29; 68% of cases occurred during December 12–18 (Figure). Antibiotics were prescribed to 36 (72%) patients. Nine (20%) ill residents were hospitalized. Two additional resident deaths occurred on December 21 and December 22, for a total of four, increasing the fatality rate to 9% among residents meeting the case definition (n = 44).
The mean age of affected residents was 81 years (range = 31–98 years); 57% were female. The most frequently reported signs and symptoms among all patients included congestion (72%), cough (60%), and fever (38%). The ill residents' rooms were distributed throughout the facility, with no apparent clustering.
Oropharyngeal swab samples were collected from 13 (30%) ill residents for respiratory viral testing by polymerase chain reaction at the Florida Department of Health Bureau of Public Health Laboratories. Ten specimens tested positive for influenza A H3, and three tested positive for respiratory syncytial virus. Among three specimens selected by the laboratory for atypical bacterial pathogens testing (Chlamydia pneumoniae, Legionella pneumophila, and Mycoplasma pneumoniae), all were negative. Urine antigen tests for L. pneumophila conducted at a commercial laboratory for 18 residents were all negative. Resident characteristics were analyzed for their association with illness including age, sex, race, room location, tobacco use, pneumococcal vaccination status, underlying chronic diseases, and obesity; no statistically significant association was found.
All asymptomatic residents and staff were considered to have been exposed, and courses of prophylactic oseltamivir were offered to exposed persons on December 21 and December 22. The facility cancelled group activities, initiated droplet precautions, and stopped accepting admissions. Additional measures included implementation of respiratory precautions for visitors and exclusion of ill staff from work until 24 hours after symptom resolution. No cases were identified after December 21.
Influenza A H3N2 was the predominant influenza virus strain circulating in the United States during the 2014–15 influenza season, and the majority of H3N2 viruses tested have drifted from the H3N2 vaccine strain (1). Three of the 10 swabs that tested positive for influenza A H3 were forwarded to CDC for further analysis; two of these samples were the nondrifted strain (A/Texas/50/2012-LIKE (H3N2) GP). Among the 44 ill residents, 19 (43%) had documentation of receipt of influenza vaccination during the 2014–15 influenza season, including two patients who were hospitalized (one of whom died). Among 51 unaffected residents, 33 (65%) had documentation of receipt of influenza vaccination.
As of January 24, 2015, widespread influenza activity and 76 reported influenza-like illness outbreaks had occurred in Florida (2), including this severe outbreak that resulted in a 46% attack rate and four deaths. The 2014–15 influenza season was moderately severe overall, especially in older adults, and reduced vaccine effectiveness was widely reported (3). Neither influenza testing nor prescription of antiviral medications occurred during the initial cluster, which was followed by extensive secondary transmission. Preventing transmission of influenza viruses within long-term care facilities requires a multifaceted approach that includes yearly vaccination of all residents and health care workers (4); prompt testing when any resident has signs and symptoms* that could be due to influenza; standard and droplet precautions for residents with suspected or confirmed influenza; empirical antiviral treatment of all residents with confirmed or suspected influenza, regardless of vaccination status; and antiviral chemoprophylaxis for residents as soon as an outbreak is identified.
Acknowledgment
Florida Department of Health, Bureau of Public Health Laboratories.
1Epidemic Intelligence Service, CDC; 2Florida Department of Health, Division of Disease Control and Health Protection; 3Florida Department of Health in Pasco County; 4Florida Department of Health, Bureau of Public Health Laboratories in Tampa.
Corresponding author: John G. Jordan, MD, jgjordan@cdc.gov, 850-245-4418.
References
- Appiah GD, Blanton L, D'Mello T, et al. Influenza activity—United States, 2014–15 season and composition of the 2015–16 influenza vaccine. MMWR Morb Mortal Wkly Rep 2015;64:583–90.
- Florida Department of Health. Florida flu review, 2014–15 season, week 3: January 18–24, 2015. Tallahassee, FL: Florida Department of Health; 2015. Available at http://www.floridahealth.gov/diseases-and-conditions/influenza/_documents/2014-15/2015_w3_flu_review.pdf.
- Flannery B, Clippard J, Zimmerman RK, et al. Early estimates of seasonal influenza vaccine effectiveness—United States, January 2015. MMWR Morb Mortal Wkly Rep 2015;64:10–5.
- LA, Shay DK, Shimabukuro TT, et al. Prevention and control of seasonal influenza with vaccines: recommendations of the Advisory Committee on Immunization Practices—United States, 2013–2014. MMWR Recomm Rep 2013;62(No. RR-07):1–43.
* Includes fever or feverishness with cough, chills, headache, myalgias, sore throat, or runny nose. Elderly patients might have atypical clinical signs and symptoms.
FIGURE. Number of cases of outbreak-associated fever or respiratory illness in nursing home residents and staff members, by date of illness onset — Florida, November–December, 2014
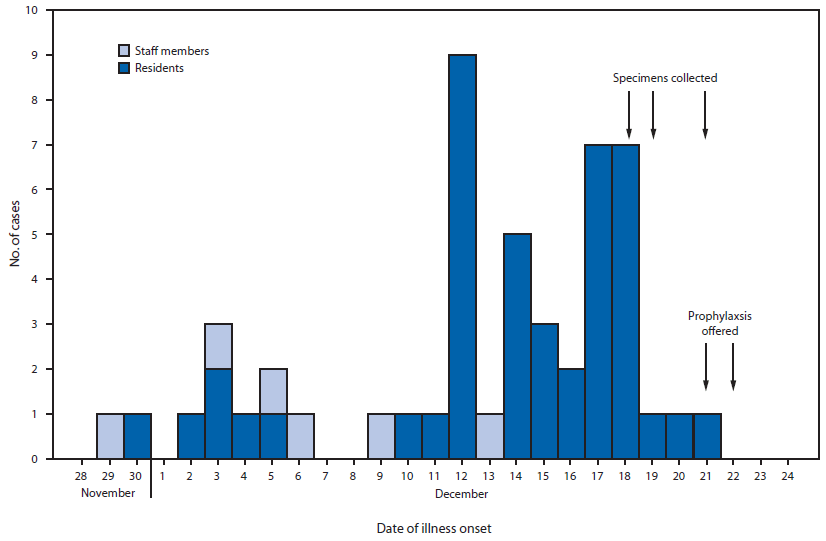
Alternate Text: The figure above is a histogram showing the number of cases of outbreak-associated fever or respiratory illness in nursing home residents and staff members, by date of illness onset, in Florida during November-December 2014.
Use of trade names and commercial sources is for identification only and does not imply endorsement by the U.S. Department of
Health and Human Services.
References to non-CDC sites on the Internet are
provided as a service to MMWR readers and do not constitute or imply
endorsement of these organizations or their programs by CDC or the U.S.
Department of Health and Human Services. CDC is not responsible for the content
of pages found at these sites. URL addresses listed in MMWR were current as of
the date of publication.
All MMWR HTML versions of articles are electronic conversions from typeset documents.
This conversion might result in character translation or format errors in the HTML version.
Users are referred to the electronic PDF version (http://www.cdc.gov/mmwr)
and/or the original MMWR paper copy for printable versions of official text, figures, and tables.
An original paper copy of this issue can be obtained from the Superintendent of Documents, U.S.
Government Printing Office (GPO), Washington, DC 20402-9371;
telephone: (202) 512-1800. Contact GPO for current prices.
**Questions or messages regarding errors in formatting should be addressed to
mmwrq@cdc.gov.
 ShareCompartir
ShareCompartir


