 |
|
|
|
|
|
|
| ||||||||||
|
|
|
|
|
|
|
||||
| ||||||||||
|
|
|
|
|
Persons using assistive technology might not be able to fully access information in this file. For assistance, please send e-mail to: mmwrq@cdc.gov. Type 508 Accommodation and the title of the report in the subject line of e-mail. Public Health and Aging: Influenza Vaccination Coverage Among Adults Aged >50 Years and Pneumococcal Vaccination Coverage Among Adults Aged >65 Years --- United States, 2002Vaccination of persons at risk for complications from influenza and pneumococcal disease is a key public health strategy in preventing morbidity and mortality in the United States. During the 1990--1999 influenza seasons, approximately 36,000 deaths were attributed annually to influenza infection, with approximately 90% of deaths occurring among adults aged >65 years (1). In 1998, an estimated 3,400 adults aged >65 years died as a result of invasive pneumococcal disease (2). One of the national health objectives for 2010 is to achieve 90% coverage of noninstitutionalized adults aged >65 years for both influenza and pneumococcal vaccinations (objective no. 14.29) (3). In 2000, the Advisory Committee on Immunization Practices (ACIP) broadened the universal recommendations for influenza vaccination to include adults aged 50--64 years in addition to adults aged >65 years. To assess progress toward achieving the 2010 national health objective and implementing the ACIP recommendations, CDC analyzed data from the 2002 Behavioral Risk Factor Surveillance System (BRFSS). This report summarizes the results of that analysis, which indicate that influenza and pneumococcal vaccination levels among adults aged >65 years and influenza vaccination levels among adults aged 50--64 years varied widely among states/areas and racial/ethnic populations. Innovative approaches are needed to increase vaccination coverage, particularly among certain populations. BRFSS is a state-based, random-digit--dialed telephone survey of the U.S. civilian, noninstitutionalized population aged >18 years. All 50 states, the District of Columbia (DC), and three U.S. territories participate in the survey. Respondents were asked, "During the past 12 months, have you had a flu shot?" and "Have you ever had a pneumonia vaccine?" For the 2002 BRFSS, the median state/area response rate was found to be 58.3% (range: 42.2%--82.6%) by using the CASRO method (4). A total of 247,964 persons responded, of whom 59,954 (24.1%) were aged 50--64 years, and 51,082 (20.6%) were aged >65 years. Respondents who reported unknown influenza (0.2%) or pneumococcal (2.7%) vaccination status were excluded from the analysis. Overall vaccination levels were estimated for the 50 states, DC, Guam, Puerto Rico, and the U.S. Virgin Islands (USVI). Data were weighted by age, sex, and, in certain states/areas, race/ethnicity to reflect the estimated adult population. SUDAAN was used to calculate point estimates and 95% confidence intervals (CIs). Multivariable logistic regression analysis was used to assess factors associated independently with receiving vaccination. In 2002, of respondents aged >65 years, 66.4% (95% CI = 65.6%--67.1%) reported having received influenza vaccine during the preceding 12 months. Vaccination coverage levels ranged from 32.2% (USVI) to 76.6% (Minnesota), with a median of 68.4% (Table 1). Among respondents aged 50--64 years, 36.4% (95% CI = 35.7%--37.1%) reported having received influenza vaccine during the preceding 12 months. Vaccination coverage levels in this age group ranged from 15.9% (Puerto Rico) to 49.0% (South Dakota), with a median of 38.4%. The proportion of respondents aged >65 years reporting ever having received pneumococcal vaccine was 61.8% (95% CI = 61.0%--62.6%). Vaccination coverage levels ranged from 25.1% (Puerto Rico) to 72.5% (North Dakota), with a median of 63.0%. Substantial variation in vaccination coverage by race/ethnicity was observed. For respondents aged >65 years, the estimated proportions of influenza and pneumococcal vaccination for non-Hispanic whites (69.0% and 64.8%, respectively) were more than those for non-Hispanic blacks (50.6% and 44.5%, respectively) and Hispanics (54.8% and 44.4%, respectively) (Table 2). Similar patterns were observed for influenza vaccination among adults aged 50--64 years: 37.9% for non-Hispanic whites, 29.8% for non-Hispanic blacks, and 29.7% for Hispanics. These differences were not explained by variations in education level, sex, self-reported health, diabetes status, asthma history, and regular source of health care. Other factors associated independently with higher receipt of influenza and pneumococcal vaccination included having a regular source of health care, having diabetes or asthma, having less than excellent/very good self-reported health, and having an education level higher than high school (Table 3). Reported by: PM Wortley, MD, Immunization Svcs Div, National Immunization Program; N Jain, MD, EIS Officer, CDC. Editorial Note:The findings in this report indicate wide variability in influenza and pneumococcal vaccination coverage across states/areas and marked differences in vaccination coverage by race/ethnicity. Influenza vaccination coverage levels among adults aged 50--64 years were <50% in all reporting areas, substantially lower than levels among adults aged >65 years. In addition, both influenza and pneumococcal vaccination levels among adults aged >65 years were substantially below the 2010 national health objective of 90% coverage. Estimates of influenza vaccination coverage among adults aged 50--64 years were low despite the revised ACIP recommendations in 2000. The universal recommendations were broadened to address the prevalence of high-risk medical conditions in adults aged 50--64 years, of whom approximately 29% have one or more chronic medical condition. Age-based strategies for vaccination have been implemented more successfully than patient-selection strategies based on medical conditions (1). Efforts are needed to increase awareness of the revised recommendations among health-care providers and the general public. Information regarding the adult immunization schedule is available at http://www.cdc.gov/nip. Factors predictive of influenza and pneumococcal vaccination were similar, and having a source for regular health care was the factor most associated with receiving either vaccination. After adjustments were made for known potential confounding factors measured by BRFSS (i.e., education level but not direct measures of access to care, which were not available), non-Hispanic whites remained more likely to be vaccinated than non-Hispanic blacks and Hispanics. Strategies for addressing these disparities are being assessed by the Racial and Ethnic Adult Disparities Immunization Initiative (READII) through a 2-year demonstration project (5). Vaccine production for the 2003--04 influenza season is proceeding on schedule, and projected production and distribution schedules will allow for a sufficient supply of influenza vaccine during October--November. Influenza vaccination may proceed for all persons at high risk and healthy persons, individually and through mass campaigns, as soon as vaccine is available and should continue until supplies are depleted. Pneumococcal vaccine should be offered all year to adults aged >65 years and other persons at high risk. The findings in this report are subject to at least three limitations. First, influenza and pneumococcal vaccination status was based on self-report and not validated. The validity of self-reported pneumococcal vaccination is lower than that of influenza vaccination (6). Second, the median BRFSS response rate (58.3%) in this survey was low. BRFSS results have been compared with results from the National Health Interview Survey (NHIS), a household-based, face-to-face interview survey with higher response rates. Comparisons show similar trends and subgroup differences; however, BRFSS vaccination estimates are consistently higher than NHIS estimates (7). Finally, because the survey is conducted during a 12-month period, questions regarding receipt of influenza vaccination do not reflect a single influenza season. The variation in influenza and pneumococcal vaccination coverage observed among states/areas suggests that opportunities exist to improve vaccination coverage. Although systems-based approaches (e.g., standing orders) have been effective in increasing vaccination coverage levels, these strategies are not implemented widely (8,9). To increase vaccination coverage levels, states/areas should promote these and other evidence-based strategies. Low provider reimbursement might be a barrier to vaccination; however, in 2003, Medicare increased its payment rates by 94% for administering influenza and pneumococcal vaccine (10). Influenza vaccination coverage among adults aged >65 years has leveled since 1997 (7), and unless substantial efforts and innovative approaches are undertaken in collaboration with public, private, and community partners, the 2010 national health objective might not be achieved. Acknowledgment This report is based on data contributed by state BRFSS coordinators. References
Table 1 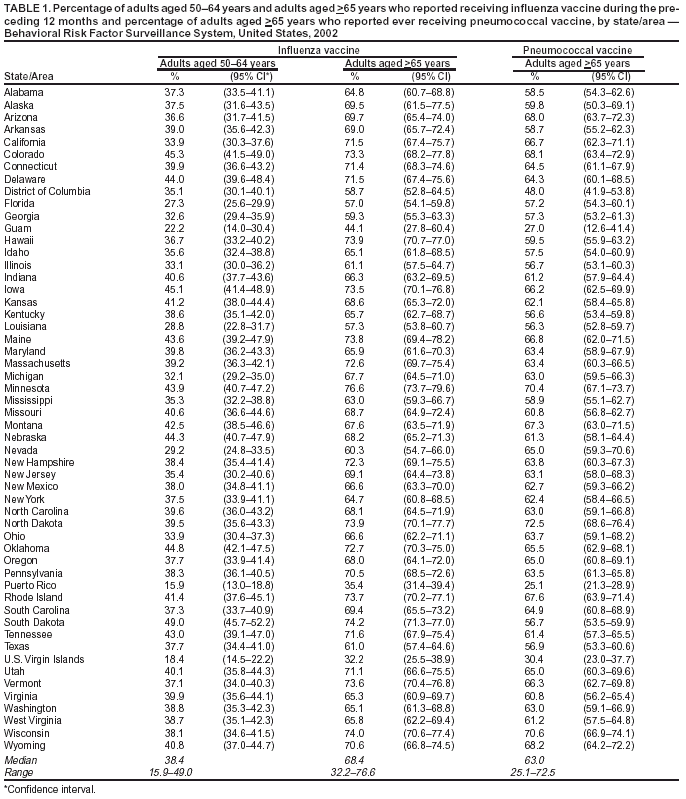 Return to top. Table 2 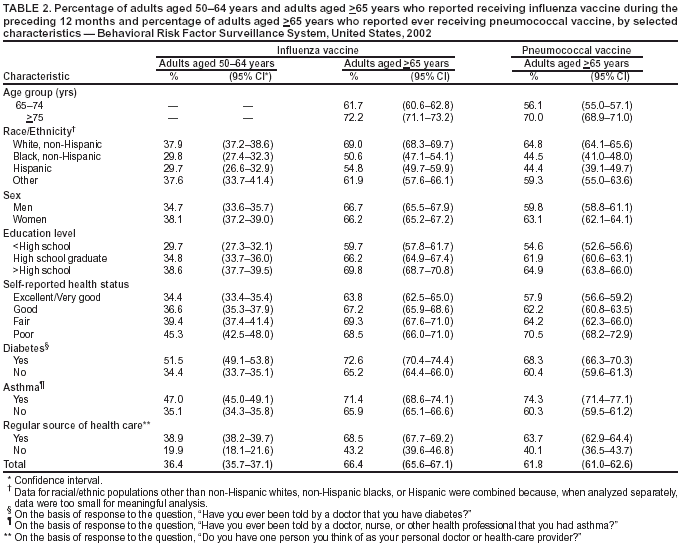 Return to top. Table 3 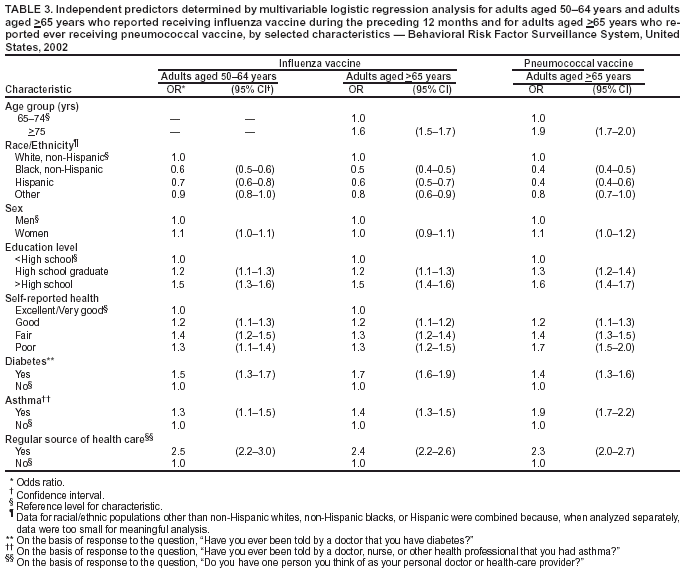 Return to top.
Disclaimer All MMWR HTML versions of articles are electronic conversions from ASCII text into HTML. This conversion may have resulted in character translation or format errors in the HTML version. Users should not rely on this HTML document, but are referred to the electronic PDF version and/or the original MMWR paper copy for the official text, figures, and tables. An original paper copy of this issue can be obtained from the Superintendent of Documents, U.S. Government Printing Office (GPO), Washington, DC 20402-9371; telephone: (202) 512-1800. Contact GPO for current prices. **Questions or messages regarding errors in formatting should be addressed to mmwrq@cdc.gov.Page converted: 10/16/2003 |
|||||||||
This page last reviewed 10/16/2003
|