 |
|
|
|
|
|
|
| ||||||||||
|
|
|
|
|
|
|
||||
| ||||||||||
|
|
|
|
|
Persons using assistive technology might not be able to fully access information in this file. For assistance, please send e-mail to: mmwrq@cdc.gov. Type 508 Accommodation and the title of the report in the subject line of e-mail. Experiences with Influenza-Like Illness and Attitudes Regarding Influenza Prevention --- United States, 2003--04 Influenza SeasonDespite advances in medical treatment, influenza results in approximately 36,000 deaths each year in the United States (1). Vaccination has been a mainstay of influenza prevention, with annual vaccination recommended for adults and children at high risk; efforts to interrupt person-to-person transmission are also important. In October 2003, CDC recommended that health-care facilities implement a Universal Respiratory Hygiene Strategy, including providing masks or facial tissues in waiting rooms to persons with respiratory symptoms (2). To gather information on influenza-like illness (ILI) and attitudes regarding prevention of ILI (including use of vaccine and respiratory hygiene), CDC and 11 Emerging Infections Programs (EIPs) conducted a random-digit--dialed telephone survey of noninstitutionalized U.S. civilian adults in February 2004. This report summarizes the results of that survey, which determined that 43% of adults and 69% of children aged 6 months--17 years with ILI visited a health-care provider for the illness. Eight percent of adults with ILI reported having been asked by a health-care provider to wear a mask; 82% said they would wear a mask if requested. With the limited availability of influenza vaccine this season, the use of masks by persons with cough illnesses in health-care settings, a component of the Universal Respiratory Hygiene Strategy, might be a helpful and acceptable method for decreasing influenza transmission. EIP is a population-based network consisting of CDC, state health departments, and local collaborators to assess the impact of emerging infections and evaluate methods for their prevention and control (3). For this survey, data were collected from a stratified random sample of telephone-equipped households in all 11 EIP surveillance areas* that covered selected counties of certain states and the entire populations of others. During February 6--22, 2004, a total of 200 eligible households in each area were selected by list-assisted random-digit--dialing from a sampling frame of possible telephone numbers filtered to eliminate unused or business exchanges. When an adult in an eligible household declined to participate or could not be contacted after six attempts, a substitute number was selected randomly from the list. The survey was confidential, and respondents gave consent for participation. One English-speaking adult aged >18 years was interviewed in each participating household. The survey assessed ILI by self-report, visits to health-care providers, prescription of medication for self-reported ILI, attitudes about vaccination against influenza, and willingness to take measures that might prevent influenza transmission. Questions addressed the period October 1, 2003, through the time of the interview in February 2004. Data were weighted to reflect the age, sex, and racial/ethnic distributions of the population of each area. Proportions, risk ratios, and confidence intervals were calculated by statistical software. A total of 2,231 surveys were completed. Among eligible respondents, 48.7% consented to participate. The median age of respondents was 43 years (range: 18--97 years), and 51.3% were female. The proportion of surveyed adults who responded "yes" to the question "Have you had an illness you thought was the flu at any time since October?" was 17.8% (Table 1). Of these, 53.2% reported having a sudden high fever with cough or sore throat. For the 811 households with at least one child aged 6 months--17 years at the time of interview, one child was randomly selected; of these, 23.9% had ILI from October 2003 through the time of the interview. Among children with reported ILI, 68.9% had symptoms of fever with cough and/or sore throat. On average, reported ILI occurrence among children and adults peaked during December 2003. Of those for whom ILI was reported, 43.3% of adults and 69.2% of children visited a health-care provider for the illness (Table 1). The provider reportedly told 64.5% of these adults and 47.6% of these children that they had influenza; 37.2% of adults with a clinical diagnosis of influenza were tested for influenza, compared with 65.0% of children. Of adults who visited a provider, 85.9% received a prescription for medication, most commonly an antibiotic (33.1%). Approximately 8.2% of adults who visited a health-care provider for ILI said they were asked to wear a mask; 82.4% of all respondents said they would wear a mask while waiting at the doctor's office or hospital if asked to by their health-care provider (Table 2). Fewer respondents agreed with the statements "people with a cough should wear a mask while waiting to see a health-care provider" and "people who are sick and able to spread germs should wear a mask in public" (62.8% and 59.0%, respectively). Approximately 70% of all respondents said they believed the influenza vaccine for the 2003--04 season was "somewhat" or "very effective," 32.8% said they believed the influenza vaccine causes influenza, and 10.7% reported experiencing problems obtaining influenza vaccine because of limited supply. Sixty percent of respondents said they planned to seek influenza vaccine during the 2004--05 influenza season, including one third of those who said they were not vaccinated during 2003--04. Among respondents from households with children, 63.8% said they would be willing to have their child or children vaccinated against influenza annually. Reported by: D Vugia, MD, California Dept of Health Svcs. K Gershman, MD, Emerging Infections Program, Colorado Dept of Public Health. JL Hadler, MD, Connecticut Dept of Public Health. S Segler, MPH, Georgia Emerging Infections Program, Veterans Affairs Medical Center and Emory Univ School of Medicine, Atlanta, Georgia. PA Ryan, MSc, Maryland Dept of Health and Mental Hygiene. R Lynfield, MD, Minnesota Dept of Health. J Baumbach, MD, New Mexico Dept of Health. NM Bennett, MD, Monroe County Health Dept, Rochester, New York. PR Cieslak, MD, Oregon Dept of Human Svcs. A Craig, MD, Tennessee Dept of Health. D Perrotta, PhD, Texas Dept of Health. C Whitney, MD, SI McCoy, MPH, E Zell, MStat, Div of Bacterial and Mycotic Diseases; D Shay, MD, Div of Viral and Rickettsial Diseases; C Rebmann, MPH, Office of Surveillance, National Center for Infectious Diseases; K Cowgill, PhD, EIS Officer, CDC. Editorial Note:The 2003--04 influenza season was notable for several reasons: 1) in certain states, the season began as early as October; 2) a suboptimal antigenic match between the dominant influenza A (H3N2) subtype circulating in the community (Fujian/411/2002) and that in the vaccine (Panama/2007/99) occurred; and 3) early reports of deaths among children helped create a high demand for vaccine (4). Because of these factors, the media focused substantial attention on influenza and vaccination. This setting provided an opportunity to evaluate the acceptability of influenza prevention measures among the public. Droplets of respiratory secretions are believed to be the primary means of person-to-person influenza transmission; spread can also occur through direct person-to-person contact or through fomites (5). For periods of increased respiratory-infection activity, the Universal Respiratory Hygiene Strategy recommends that health-care facilities provide masks or facial tissues to persons who are coughing and that hand-hygiene agents (e.g., alcohol-based hand gels) be readily available (2). Use of surgical masks by health-care professionals to protect against infection with influenza and other respiratory pathogens is also an established component of hospital infection control (6); however, its effectiveness in preventing transmission from influenza patients to others in outpatient settings has not been determined (4). Hand washing has been demonstrated to be effective in reducing respiratory illness, and alcohol-based hand sanitizers can kill influenza viruses on hands; however, studies of hand-hygiene measures on influenza transmission are lacking (5,7). Likewise, few published data are available on the public's willingness to wear masks or use other measures to prevent transmission of respiratory illnesses. The survey results indicated that at least 8% of respondents with ILI had been asked by their health-care providers to wear a mask while waiting to be examined, and more than 80% of respondents indicated a willingness to do so in the future. Information about the acceptability of such intervention measures might be useful in managing large outbreaks, including pandemic influenza. Although only 8% of those visiting a health-care provider for ILI were asked to wear a mask, many of those visits might have been to doctors' offices; mask usage might be higher in other health-care settings. Use of tissues for reducing droplet spread and use of hand-hygiene agents were not evaluated in this survey. With the limited availability of influenza vaccine this season, the use of masks by persons with cough illnesses in health-care settings and other components of the Universal Respiratory Hygiene Strategy (e.g., tissues and alcohol-based hand sanitizers) might help decrease influenza transmission. The survey findings suggest that children were disproportionately affected by ILI during the 2003--04 season. Reported ILI was one third more common among children than adults, and reported symptoms in children were more frequently consistent with the ILI surveillance case definition (temperature >100.0°F [>37.8°C] and cough or sore throat in the absence of a known cause other than influenza). Health-care use (including provider visits, influenza testing, and hospitalization) was more common among children than adults with reported ILI. Recent recommendations for providing influenza vaccine to all young children were designed to address the substantial disease burden among this age group (1). More than one half of respondents from households with children said they would participate in annual influenza vaccination of children. The findings in this report are subject to at least two limitations. First, certain sampling factors (low response rate, limited sampling area, and restriction to English-speaking respondents) might mean that some of the results are not representative of the entire U.S. population. Second, self-reported ILI symptoms are not specific for influenza; because other etiologic agents can cause influenza-like symptoms, the true incidence of influenza is expected to be lower (8). In addition to indicating that persons with respiratory illness might be willing to wear masks in health-care settings, the results of this survey also suggest opportunities for improving vaccination coverage. Although a majority of respondents said they believed the 2003--04 influenza vaccine was "somewhat" or "very effective," at least one tenth reported problems obtaining vaccine, and nearly one third reported believing that influenza vaccine causes influenza. Educational efforts about the effectiveness of influenza vaccination and improved supply and distribution of vaccine might improve vaccination coverage levels. Acknowledgments The findings in this report are based, in part, on contributions by ORC/MACRO, Inc., Washington, DC. S Feavor, MPH, C Miller, MPH, R Danila, PhD, Minnesota Dept of Health. W Schaffner, MD, B Barnes, Vanderbilt Univ School of Medicine, Nashville, Tennessee. ML Cartter, MD, Connecticut Dept of Public Health. A Thomas, MD, Oregon Dept of Human Svcs. R Pinner, MD, A Slaughter, Office of the Director; A Banerjee, Div of Bacterial and Mycotic Diseases, National Center for Infectious Diseases, CDC. References
* EIP surveillance areas include the entire states of Connecticut, Georgia, Maryland, Minnesota, New Mexico, Oregon, Tennessee, and Texas, and selected counties in California (Alameda, Contra Costa, and San Francisco), Colorado (Adams, Arapahoe, Boulder, Broomfield, Denver, Douglas, and Jefferson), New York (Albany, Columbia, Erie, Genesee, Greene, Livingston, Monroe, Montgomery, Niagara, Ontario, Orleans, Rensselaer, Saratoga, Schenectady, Schoharie, Wayne, Wyoming, and Yates), and Tennessee (Cheatham, Davidson, Dickson, Hamilton, Knox, Robertson, Rutherford, Shelby, Sumner, Williamson, and Wilson). Table 1 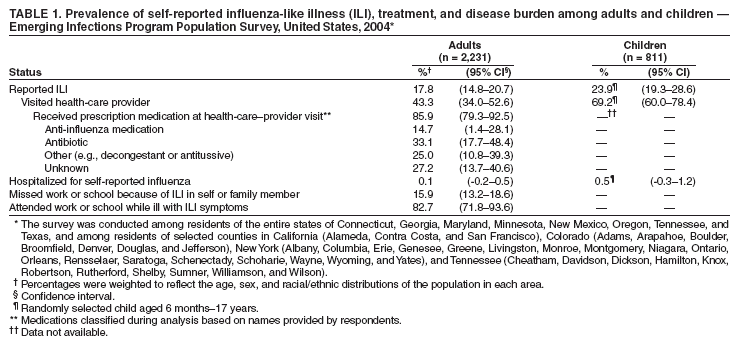 Return to top. Table 2  Return to top.
Disclaimer All MMWR HTML versions of articles are electronic conversions from ASCII text into HTML. This conversion may have resulted in character translation or format errors in the HTML version. Users should not rely on this HTML document, but are referred to the electronic PDF version and/or the original MMWR paper copy for the official text, figures, and tables. An original paper copy of this issue can be obtained from the Superintendent of Documents, U.S. Government Printing Office (GPO), Washington, DC 20402-9371; telephone: (202) 512-1800. Contact GPO for current prices. **Questions or messages regarding errors in formatting should be addressed to mmwrq@cdc.gov.Page converted: 12/16/2004 |
|||||||||
This page last reviewed 12/16/2004
|