Persons using assistive technology might not be able to fully access information in this file. For assistance, please send e-mail to: mmwrq@cdc.gov. Type 508 Accommodation and the title of the report in the subject line of e-mail.
Update: Influenza Activity --- United States, August 30--October 31, 2009
The 2009 pandemic influenza A (H1N1) virus emerged in the United States in April 2009 (1) and has since spread worldwide. Influenza activity resulting from this virus occurred throughout the summer and, by late August, activity had begun to increase in the southeastern United States (2). Since August, activity has increased in all regions of the United States. As of the week ending October 31, nearly all states were reporting widespread disease. Since April 2009, pandemic H1N1 has remained the dominant circulating influenza strain. This report summarizes U.S. influenza activity* from August 30, 2009, defined as the beginning of the 2009--10 influenza season, through October 31, 2009.
Viral Surveillance
During August 30--October 31, World Health Organization (WHO) and National Respiratory and Enteric Virus Surveillance System (NREVSS) collaborating laboratories in the United States tested 163,123 respiratory specimens for influenza viruses, 48,585 (30%) of which were positive (Figure 1). Of the 48,483 (99.8%) specimens positive for influenza A, 32,867 (68%) were subtyped by real-time reverse transcription--polymerase chain reaction (rRT-PCR) or by virus culture. A total of 32,814 (99.8%) of these were 2009 pandemic influenza A (H1N1) viruses, 18 (0.1%) were seasonal influenza A (H1), and 35 (0.1%) were influenza A (H3) viruses.
CDC has antigenically characterized 239 pandemic influenza A (H1N1)viruses collected since September 1. A total of 238 (99.6%) of the 239 pandemic influenza A (H1N1) viruses tested were antigenically related to the A/California/7/2009 (H1N1)pdm reference virus selected by WHO as the 2009 pandemic influenza A (H1N1) vaccine virus; one virus (0.4%) tested showed reduced titers with antisera produced against A/California/7/2009.
Antiviral Resistance of Influenza Virus Isolates
CDC conducts surveillance for resistance of circulating influenza viruses to influenza antiviral medications: adamantanes (amantadine and rimantadine) and neuraminidase inhibitors (zanamivir and oseltamivir). Since September 1, a total of 256 pandemic influenza A (H1N1) virus isolates collected in the United States have been tested for resistance to the neuraminidase inhibitors. All but four were susceptible to oseltamivir, bringing the total number of such resistant isolates to 14 since April 2009. Twelve of the 14 patients from whom the resistant isolates were collected had documented exposure to oseltamivir through treatment or chemoprophylaxis. Exposure to oseltamivir has yet to be determined for one patient, and another patient had no documented oseltamivir exposure. All 256 tested viruses were sensitive to the neuraminidase inhibitor zanamivir. Since September 1, one influenza A (H3N2) virus isolate and 152 pandemic influenza A (H1N1) virus isolates also have been tested for resistance to adamantanes (amantadine and rimantadine); all of these virus isolates were resistant to the adamantanes.
State-Specific Activity Levels
During the first week of the influenza season (August 30--September 5), 11 states, clustered mainly in the South, reported widespread activity. By the following week, that number had more than doubled to 26 states. In subsequent weeks, more states reported increased activity. As of the week ending October 31, widespread influenza activity† was reported by all but two states (Mississippi and Hawaii). In contrast, during the 2008--09 influenza season, no state reported widespread influenza activity before the week ending January 10, 2009.
Outpatient Illness Surveillance
The weekly percentage of outpatient visits for influenza-like illness (ILI)§ reported by the U.S. Outpatient ILI Surveillance Network (ILINet) increased from 3.5% in the week ending September 5 to 7.7% in the week ending October 31 (Figure 2). ILI activity has remained above the national baseline of 2.3% during this entire period.¶ Since the week ending October 3, all 10 surveillance regions have reported a percentage of outpatient visits for ILI at or above their region-specific baseline levels. These percentages are all substantially elevated compared with data recorded in previous years over the same period.
Influenza-Associated Hospitalizations
Laboratory-confirmed influenza-associated hospitalizations are monitored using a population-based surveillance network that includes the 10 Emerging Infections Program (EIP) sites and six new sites.** During September--October, cumulative influenza hospitalization rates for persons aged <65 years were substantially elevated for this time of year and exceeded or were approaching the end-of-season cumulative rates for the last three seasons. Preliminary cumulative rates of laboratory-confirmed, influenza-associated hospitalizations reported for children aged 0--4 years were 3.1 per 10,000 population by EIP and 7.3 per 10,000 population by the new sites (Figure 3). Rates for other age groups were as follows: 5--17 years, 1.5 by EIP and 2.9 by the new sites; 18--49 years, 1.2 by EIP and 1.2 by the new sites; 50--64 years, 1.3 by EIP and 1.2 by the new sites; and >65 years, 1.0 by EIP and 1.1 by the new sites.
On August 30, CDC and the Council of State and Territorial Epidemiologists (CSTE) instituted modified case definitions for aggregate reporting of influenza-associated hospitalizations and deaths. This cumulative state-level reporting is referred to as the Aggregate Hospitalization and Death Reporting Activity (AHDRA).†† During August 30--October 31, a total of 17,838 hospitalizations associated with laboratory-confirmed influenza virus infections were reported to CDC through AHDRA. On average, 31 states each week reported laboratory-confirmed hospitalizations during that period.
Pneumonia- and Influenza-Related Mortality
Influenza-associated deaths are monitored by the 122 Cities Mortality Reporting System and AHDRA. For the week ending October 31, pneumonia or influenza was reported as an underlying or contributing cause of death for 7.4% of all deaths reported through the 122 Cities Mortality Reporting System, above the week-specific epidemic threshold of 6.7%§§ and the fifth consecutive week above the epidemic threshold.
During August 30--October 31, 672 deaths associated with laboratory-confirmed influenza virus infections were reported to CDC through AHDRA. On average, 29 states reported laboratory-confirmed deaths each week during that period. The 672 laboratory-confirmed deaths are in addition to the 593 laboratory-confirmed deaths from 2009 pandemic influenza A (H1N1) that were reported to CDC from April through August 30, 2009.
Influenza-Associated Pediatric Mortality
During August 30--October 31, CDC received 85 reports of pediatric deaths associated with influenza infection (Figure 4). Seventy-three of these cases were associated with laboratory-confirmed 2009 pandemic influenza A (H1N1) virus. The remaining 12 pediatric deaths were associated with an influenza A infection for which the subtype was undetermined.
Of the 85 pediatric deaths reported since August 30, a total of 12 (14%) were among children aged <2 years, nine (11%) were among children aged 2--4 years, 30 (35%) were among children aged 5--11 years, and 34 (40%) were among children aged 12--17 years. Seventy-eight (92%) of the 85 decedents had a medical history reported. Of the 78, 56 (72%) had one or more medical conditions associated with an increased risk for influenza-related complications (3).
Since April 26, CDC has received 145 reports of pediatric deaths associated with influenza infection. Of these, 129 (89%) cases were associated with laboratory-confirmed 2009 pandemic influenza A (H1N1) virus. The remaining 16 pediatric deaths were associated with seasonal influenza or an influenza A virus for which the subtype was undetermined. In comparison, during the preceding five influenza seasons, the total number of reported pediatric influenza deaths ranged from 46 to 153, with an average of 82 deaths each year.
Reported by: WHO Collaborating Center for Surveillance, Epidemiology, and Control of Influenza. L Brammer, MPH, S Epperson, MPH, L Blanton, MPH, R Dhara, MPH, T Wallis, MS, L Finelli, DrPH, T Fiore, MD, L Gubareva, PhD, J Bresee, MD, L Kamimoto, MD, X Xu, MD, A Klimov, PhD, C Bridges, MD, N Cox, PhD, Influenza Div, National Center for Immunization and Respiratory Diseases, CDC; C Cox, MD, EIS Officer, CDC.
Editorial Note:
During August 30--October 31, influenza activity was substantially above historic levels in all U.S. surveillance systems. By mid-October, nearly all states reported geographically widespread influenza activity. Nationwide, the percentage of visits to health-care providers for ILI was higher than that observed at the peak of any seasonal influenza season since ILINet was implemented in its current form in 1997. Influenza-associated hospitalization rates continued to trend upward in all age groups, substantially above historical rates from the same time period during previous years. The widespread occurrence of pandemic H1N1 influenza in the United States highlights the importance of understanding and appropriately using available tools for prevention and treatment of influenza. Particularly important in reducing the impact of pandemic H1N1 infections are recommendations for the use of influenza A (H1N1) 2009 monovalent vaccines and a continued emphasis on early, empiric antiviral treatment of hospitalized patients and others who are ill and at greater risk for influenza-related complications.
Severe outcomes among children, continue to be prominent during the 2009 influenza A (H1N1) pandemic. A total of 145 pediatric deaths associated with influenza infection have been reported since April 26. In comparison, 82 deaths were reported on average during the previous five influenza seasons. Pediatric hospitalization rates are higher than those of any other age group and are particularly high among children aged <5 years. These epidemiologic data provide support for ACIP recommendations that include persons aged 6 months-24 years in the initial target groups for vaccination using the influenza A (H1N1) 2009 monovalent vaccine now available (3). In addition, vaccination providers should vaccinate persons who live with or care for infants aged <6 months because young infants themselves cannot be vaccinated. Other target groups for initial supplies of influenza A (H1N1) 2009 monovalent vaccine include pregnant women, health-care and emergency medical services personnel, and persons aged 25--64 years who are at higher risk for more severe disease because of chronic health disorders or compromised immune systems (3).
The supply of influenza A (H1N1) 2009 monovalent vaccines will continue to increase rapidly through November and December.¶¶ However, these vaccines are not yet available to all persons who might benefit from vaccination. In the absence of widespread immunity based on vaccination, early empiric antiviral treatment of persons who are severely ill or at high risk for influenza-related complications can reduce the number of severe illnesses from pandemic H1N1. Observational studies of hospitalized patients with seasonal influenza and pandemic H1N1 influenza have suggested that mortality is reduced among hospitalized patients who received antiviral medications (4--5). However, the use of antiviral treatment for hospitalized patients remains suboptimal, as highlighted in recent studies indicating that 21%--25% of hospitalized patients with laboratory-confirmed pandemic H1N1 did not receive antiviral medications and, among those who did, treatment was often delayed until 1--2 days after admission (6--7). Antiviral medications active against influenza are widely available, and early empiric treatment with oseltamivir or zanamivir of hospitalized persons and others who are severely ill or at high risk for influenza-related complications is recommended (8). In addition, peramivir, an investigational intravenous neuraminidase inhibitor medication, has recently been made available under an Emergency Use Authorization by the Food and Drug Administration. Peramivir is available for treatment of certain adult and pediatric patients with suspected or laboratory-confirmed pandemic H1N1 (9,10).
The current dominant influenza virus by far is 2009 pandemic influenza A (H1N1); seasonal influenza viruses continue to circulate at low levels in the United States and elsewhere. However, influenza circulation patterns remain unpredictable, and seasonal influenza viruses might circulate more widely later in the influenza season. CDC will continue to monitor changes in geographic spread, type, and severity of 2009 pandemic influenza A (H1N1) and will issue weekly online FluView reports.*** Additional detailed information regarding 2009 pandemic influenza A (H1N1) also is available online.†††
Acknowledgments
This report is based, in part, on data contributed by participating state and territorial health departments and state public health laboratories, World Health Organization collaborating laboratories, National Respiratory and Enteric Virus Surveillance System collaborating laboratories, the U.S. Outpatient ILI Surveillance Network, the Emerging Infections Program, the Aggregate Hospitalization and Death Reporting Activity, the Influenza Associated Pediatric Mortality Surveillance System, and the 122 Cities Mortality Reporting System.
References
- CDC. Swine influenza A (H1N1) infection in two children---southern California, March--April 2009. MMWR 2009;58:400--2.
- CDC. Update: influenza activity---United States, April--August 2009. MMWR 2009;58:1009--12.
- CDC. Use of influenza A (H1N1) 2009 monovalent vaccine: recommendations of the Advisory Committee on Immunization Practices (ACIP), 2009. MMWR 2009;58(No. RR-10).
- McGeer A, Green KA, Plevneshi A, et al. Antiviral therapy and outcomes of influenza requiring hospitalization in Ontario, Canada. Clin Infect Dis 2007;45:1568--75.
- Domínguez-Cherit G, Lapinsky SE, Macias AE, et al. Critically ill patients with 2009 influenza A(H1N1) in Mexico. JAMA 2009 [Epub ahead of print].
- Louie JK, Acosta M, Winter K, et al. Factors associated with death or hospitalization due to pandemic 2009 influenza A(H1N1) infection in California. JAMA 2009;302:1896--902.
- Jain S, Kamimoto L, Bramley AM, et al. Hospitalized patients with 2009 H1N1 influenza in the United States, April--June 2009. N Eng J Med 2009 [Epub ahead of print].
- CDC. Updated interim recommendations for the use of antiviral medications in the treatment and prevention of influenza for the 2009--2010 season. Atlanta, GA: CDC; October 16, 2009. Available at http://www.cdc.gov/h1n1flu/recommendations.htm. Accessed November 9, 2009.
- CDC. Antiviral treatment options, including intravenous peramivir, for treatment of influenza in hospitalized patients for the 2009--2010 season. October 26, 2009. Available at http://www.cdc.gov/h1n1flu/eua/peramivir_recommendations.htm. Accessed November 9, 2009.
- Birnkrant D, Cox E. The emergency use authorization of peramivir for treatment of 2009 H1N1 influenza. N Engl J Med 2009. [Epub ahead of print].
* The CDC influenza surveillance system collects five categories of information from eight data sources: 1) viral surveillance (World Health Organization collaborating U.S. laboratories, the National Respiratory and Enteric Virus Surveillance System, and novel influenza A virus case reporting), 2) outpatient illness surveillance (U.S. Outpatient ILI Surveillance Network), 3) mortality (122 Cities Mortality Reporting System and influenza-associated pediatric mortality reports), 4) hospitalizations (Emerging Infections Program) and 5) summary of geographic spread of influenza (state and territorial epidemiologist reports).
† Levels of activity are 1) no activity; 2) sporadic: isolated laboratory-confirmed influenza cases or a laboratory-confirmed outbreak in one institution, with no increase in influenza-like illness (ILI) activity; 3) local: increased ILI, or at least two institutional outbreaks (ILI or laboratory-confirmed influenza) in one region with recent laboratory evidence of influenza in that region; virus activity no greater than sporadic in other regions; 4) regional: increased ILI activity or institutional outbreaks (ILI or laboratory-confirmed influenza) in at least two but less than half of the regions in the state with recent laboratory evidence of influenza in those regions; and 5) widespread: increased ILI activity or institutional outbreaks (ILI or laboratory-confirmed influenza) in at least half the regions in the state with recent laboratory evidence of influenza in the state.
§ Defined as a temperature of ≥100.0°F (≥37.8°C), oral or equivalent, and cough and/or sore throat, in the absence of a known cause other than influenza.
¶ The national and regional baselines are the mean percentage of visits for ILI during noninfluenza weeks for the previous three seasons plus two standard deviations. A noninfluenza week is a week during which <10% of specimens tested positive for influenza. National and regional percentages of patient visits for ILI are weighted on the basis of state population. Use of the national baseline for regional data is not appropriate. Additional information available at http://www.cdc.gov/flu/weekly/fluactivity.htm.
** EIP currently conducts surveillance for laboratory-confirmed, influenza-related hospitalizations in 61 counties and Baltimore, Maryland. The EIP catchment area includes 13 metropolitan areas located in 10 states. Beginning in September 2009, six new EIP sites covering 40 counties began reporting influenza-related hospitalization surveillance. Hospital laboratory, admission, and discharge databases, and infection-control logs are reviewed to identify persons with a positive influenza test (i.e., viral culture, direct fluorescent antibody assays, rRT-PCR, serology, or a commercial rapid antigen test) from testing conducted as part of their routine care.
†† States report weekly to CDC either 1) laboratory-confirmed influenza hospitalizations and deaths or 2) pneumonia and influenza syndrome--based cases of hospitalization and death resulting from all types or subtypes of influenza. Although only the laboratory-confirmed cases are included in this report, CDC continues to analyze data both from laboratory-confirmed and syndromic hospitalizations and deaths.
§§ The seasonal baseline proportion of pneumonia and influenza deaths is projected using a robust regression procedure in which a periodic regression model is applied to the observed percentage of deaths from pneumonia and influenza that were reported by the 122 Cities Mortality Reporting System during the preceding 5 years. The epidemic threshold is 1.645 standard deviations above the seasonal baseline.
¶¶ Available at http://www.cdc.gov/h1n1flu/vaccination/vaccinesupply.htm.
*** Available at http://www.cdc.gov/flu/weekly.
††† Available at http://www.cdc.gov/h1n1flu.
|
What is already known on this topic? The 2009 pandemic influenza A (H1N1) virus emerged in the United States in April 2009 and continues to cause significant disease. What is added by this report? Pediatric hospitalization rates related to pandemic H1N1 are higher than all other age groups, and influenza-related pediatric deaths continue to rise. What are the implications for public health practice? Current epidemiologic data support key actions by public health agencies, including vigorous vaccination campaigns for target groups recommended by Advisory Committee on Immunization Practices (ACIP), especially persons aged 6 months--24 years; 2) early empiric antiviral treatment of hospitalized persons and others who are severely ill or at high risk for influenza-related complications; and 3) continued emphasis of nonpharmaceutical strategies to limit the spread of influenza, such as frequent hand washing and staying home when ill. |
FIGURE 1. Number and percentage of respiratory specimens testing positive for influenza reported by World Health Organization and National Respiratory and Enteric Virus Surveillance System collaborating laboratories, by type and subtype, and surveillance week --- United States, 2009--10 influenza season
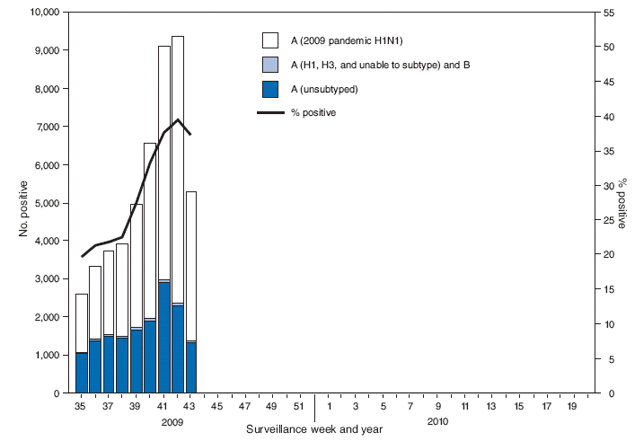
Alternate Text: The figure above shows the number and percentage of respiratory specimens testing positive for influenza reported by World Health Organization and National Respiratory and Enteric Virus Surveillance System collaborating laboratories, by type, and surveillance week for the United States, during the 2009-10 influenza season.
FIGURE 2. Percentage of outpatient visits for influenza-like illness (ILI),* by surveillance week --- U.S. Outpatient Influenza-Like Illness Surveillance Network (ILINet), United States, 2009--10, 2008--09, and 2007--08 influenza seasons
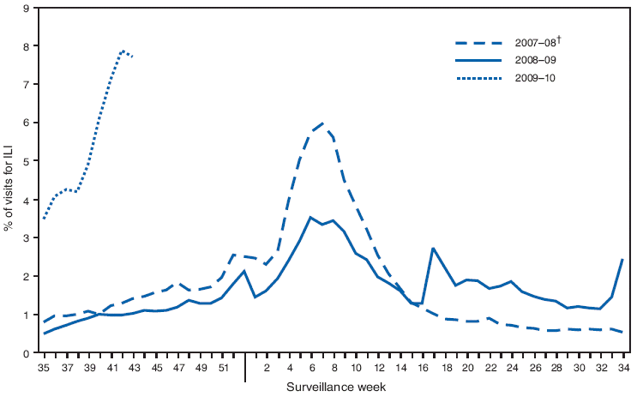
* Defined as a temperature of ≥100.0°F (≥37.8°C), oral or equivalent, and cough and/or sore throat, in the absence of a known cause other than influenza.
† No week 53 occurred during the 2007--08 influenza season; therefore, the week 53 data point for that season is an average of weeks 52 and 1.
Alternate Text: The figure shows the percentage of outpatient visits for influenza-like illness in the United States by surveillance week during the 2009-09, 2007-08 and 2007-08 influenza seasons from the U.S. Outpatient Influenza-Like Illness Surveillance Network (ILINet). The weekly percentage of outpatient visits increased to 7.7% in the week ending October 31. ILI activity has remained above the national baseline during this entire period. Since the week ending October 3, all 10 surveillance regions have reported a percentage of outpatient visits for ILI above their region-specific baseline levels. These percentages are all substantially elevated compared with data recorded in previous years over the same period.
FIGURE 3. Laboratory-confirmed influenza hospitalization rates per 10,000 population, by age group and surveillance week --- Emerging Infections Program (EIP), United States, 2009--10, 2008--09, 2007--08, and 2006--07 influenza seasons
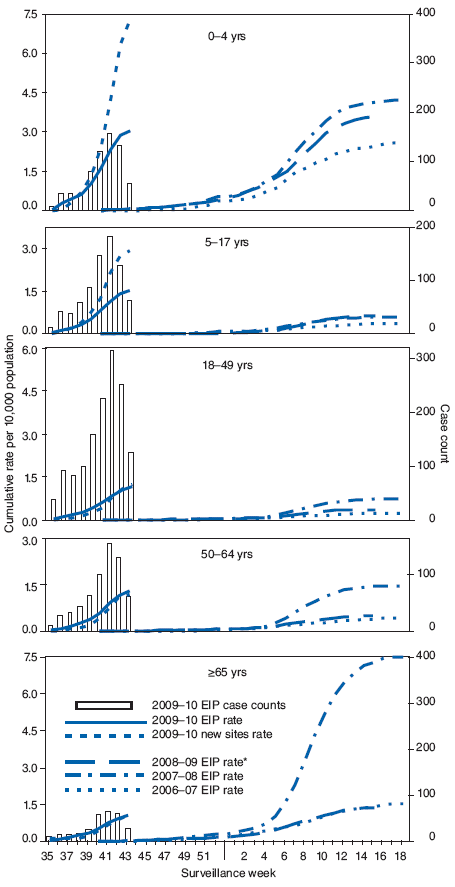
* The 2008--09 EIP rate ended as of April 14, 2009, with onset of the 2009 pandemic influenza A (H1N1) season.
Alternate Text: The figure above shows laboratory-confirmed influenza hospitalization rates per 10,000 population, by age group and surveillance week in the United States, from the Emerging Infections Program (EIP), for the 2009-10, 2008-09, 2007-08, and 2006-07 influenza seasons. During September-October, cumulative influenza hospitalization rates for persons aged <65 years were substantially elevated for this time of year and exceeded or were approaching the end-of-season cumulative rates for the last three seasons. Preliminary cumulative rates of laboratory confirmed, influenza-associated hospitalizations reported for children aged 0-4 years were 3.1 per 10,000 population by EIP and 7.3 per 10,000 population by the new sites. Rates for other age groups were as follows: 5-17 years, 1.5 by EIP and 2.9 by the new sites; 18-49 years, 1.2 by EIP and 1.2 by the new sites; 50-64 years, 1.3 by EIP and 1.2 by the new sites; and >65 years, 1.0 by EIP and 1.1 by the new sites.
FIGURE 4. Number of influenza-associated pediatric deaths, by week of death and influenza status --- United States, 2009--10, 2008--09, 2007--08, and 2006--07 influenza seasons
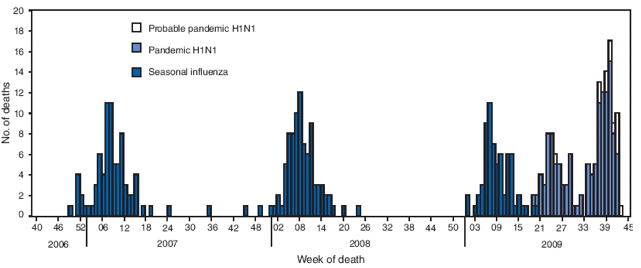
Alternate Text: The figure above shows the number of influenza-associated pediatric deaths, by week of death and influenza status in the United States from the 2009-10, 2008-09, 2007-08, and 2006-07 influenza seasons. During August 30-October 31, 2009, CDC received 85 reports of pediatric deaths associated with influenza infection.
Use of trade names and commercial sources is for identification only and does not imply endorsement by the U.S. Department of
Health and Human Services. |
All MMWR HTML versions of articles are electronic conversions from typeset documents. This conversion might result in character translation or format errors in the HTML version. Users are referred to the electronic PDF version (http://www.cdc.gov/mmwr) and/or the original MMWR paper copy for printable versions of official text, figures, and tables. An original paper copy of this issue can be obtained from the Superintendent of Documents, U.S. Government Printing Office (GPO), Washington, DC 20402-9371; telephone: (202) 512-1800. Contact GPO for current prices.
**Questions or messages regarding errors in formatting should be addressed to mmwrq@cdc.gov.Date last reviewed: 11/12/2009


