Hepatitis A
On this Page
Hepatitis A
- Epidemic jaundice attributed to Hippocrates
- Differentiated from hepatitis B in 1940s
- Serologic tests developed in 1970s
- Vaccines licensed in 1995 and 1996
The first descriptions of hepatitis (epidemic jaundice) are generally attributed to Hippocrates. Outbreaks of jaundice, probably hepatitis A, were reported in the 17th and 18th centuries, particularly in association with military campaigns. Hepatitis A (formerly called infectious hepatitis) was first differentiated epidemiologically from hepatitis B, which has a longer incubation period, in the 1940s. Development of serologic tests allowed definitive diagnosis of hepatitis B. In the 1970s, identification of the virus, and development of serologic tests helped differentiate hepatitis A from other types of non-B hepatitis.
Until 2004, hepatitis A was the most frequently reported type of hepatitis in the United States. In the prevaccine era, the primary methods used for preventing hepatitis A were hygienic measures and passive protection with immune globulin (IG). Hepatitis A vaccines were licensed in 1995 and 1996. These vaccines provide long-term protection against hepatitis A virus (HAV) infection. The similarities between the epidemiology of hepatitis A and poliomyelitis suggest that widespread vaccination of appropriate susceptible populations can substantially lower disease incidence, eliminate virus transmission, and ultimately, eliminate HAV infection.
Hepatitis A Virus
- Picornavirus (RNA)
- Humans are only natural host
- Stable at low pH
- Inactivated by temperature of 185°F or higher, formalin, chlorine
Hepatitis A Virus
Hepatitis A is caused by infection with HAV, a nonenveloped RNA virus that is classified as a picornavirus. It was first isolated in 1979. Humans are the only natural host, although several nonhuman primates have been infected in laboratory conditions. Depending on conditions, HAV can be stable in the environment for months. The virus is relatively stable at low pH levels and moderate temperatures but can be inactivated by high temperature (185°F [85°C] or higher), formalin, and chlorine.
Hepatitis A Pathogenesis
- Entry into mouth
- Viral replication in the liver
- Virus present in blood and feces 10-12 days after infection
- Virus excretion may continue for up to 3 weeks after onset of symptoms
Pathogenesis
HAV is acquired by mouth (through fecal-oral transmission) and replicates in the liver. After 10-12 days, virus is present in blood and is excreted via the biliary system into the feces. Peak titers occur during the 2 weeks before onset of illness. Although virus is present in serum, its concentration is several orders of magnitude less than in feces. Virus excretion begins to decline at the onset of clinical illness, and has decreased significantly by 7-10 days after onset of symptoms. Most infected persons no longer excrete virus in the feces by the third week of illness. Children may excrete virus longer than adults.
Hepatitis A Clinical Features
- Incubation period 28 days (range 15-50 days)
- Illness not specific for hepatitis A
- Likelihood of symptomatic illness directly related to age
- Children generally asymptomatic, adults symptomatic
Clinical Features
The incubation period of hepatitis A is approximately 28 days (range 15-50 days). The clinical course of acute hepatitis A is indistinguishable from that of other types of acute viral hepatitis. The illness typically has an abrupt onset of fever, malaise, anorexia, nausea, abdominal discomfort, dark urine and jaundice. Clinical illness usually does not last longer than 2 months, although 10%-15% of persons have prolonged or relapsing signs and symptoms for up to 6 months. Virus may be excreted during a relapse.
The likelihood of symptomatic illness from HAV infection is directly related to age. In children younger than 6 years of age, most (70%) infections are asymptomatic. In older children and adults, infection is usually symptomatic, with jaundice occurring in more than 70% of patients.
Complications
Severe clinical manifestations of hepatitis A infection are rare, however atypical complications may occur, including immunologic, neurologic, hematologic, pancreatic, and renal extrahepatic manifestations. Relapsing hepatitis, cholestatic hepatitis A, hepatitis A triggering autoimmune hepatitis, subfulminant hepatitis, and fulminant hepatitis have also been reported. Fulminant hepatitis is the most severe rare complication, with mortality estimates up to 80%. In the prevaccine era, fulminant hepatitis A caused about 100 deaths per year in the United States. The hepatitis A case-fatality rate among persons of all ages with reported cases was approximately 0.3% but may have been higher among older persons (approximately 2% among persons 40 years of age and older) More recent case-fatality estimates range from 0.3%-0.6% for all ages and up to 1.8% among adults aged >50 years. Vaccination of high risk groups and public health measures have significantly reduced the number of overall hepatitis A cases and fulminant HAV cases. Nonetheless, hepatitis A results in substantial morbidity, with associated costs caused by medical care and work loss.
Laboratory Diagnosis
Hepatitis A cannot be distinguished from other types of viral hepatitis on the basis of clinical or epidemiologic features alone. Serologic testing is required to confirm the diagnosis. Virtually all patients with acute hepatitis A have detectable IgM anti-HAV. Acute HAV infection is confirmed during the acute or early convalescent phase of infection by the presence of IgM anti-HAV in serum. IgM generally becomes detectable 5-10 days before the onset of symptoms and can persist for up to 6 months.
IgG anti-HAV appears in the convalescent phase of infection, remains present in serum for the lifetime of the person, and confers enduring protection against disease. The antibody test for total anti-HAV measures both IgG anti-HAV and IgM anti-HAV. Persons who are total anti-HAV positive and IgM anti-HAV negative have serologic markers indicating immunity consistent with either past infection or vaccination.
Molecular virology methods such as polymerase chain reaction (PCR)-based assays can be used to amplify and sequence viral genomes. These assays are helpful to investigate common-source outbreaks of hepatitis A. Providers with questions about molecular virology methods should consult with their state health department or the CDC Division of Viral Hepatitis.
Medical Management
There is no specific treatment for hepatitis A virus infection. Treatment and management of HAV infection are supportive.
Epidemiology
Hepatitis A Epidemiology
- Reservoir
- human
- Transmission
- fecal-oral
- Temporal pattern
- none
- Communicability
- 2 weeks before illness to 1 week after onset of jaundice
Occurrence
Hepatitis A occurs throughout the world. It is highly endemic in some areas, particularly Central and South America, Africa, the Middle East, Asia, and the Western Pacific.
Reservoir
Humans are the only natural reservoir of the virus. There are no insect or animal vectors. A chronic HAV state has not been reported.
Transmission
HAV infection is acquired primarily by the fecal-oral route by either person-to-person contact or ingestion of contaminated food or water. Since the virus is present in blood during the illness prodrome, HAV has been transmitted on rare occasions by transfusion. Although HAV may be present in saliva, transmission by saliva has not been demonstrated. Waterborne outbreaks are infrequent and are usually associated with sewage-contaminated or inadequately treated water.
Temporal Pattern
There is no appreciable seasonal variation in hepatitis A incidence. In the prevaccine era, cyclic increases in reported acute cases were observed every 5- 10 years, and were characterized by large community outbreaks of disease. Since introduction of vaccination in the United States, these increases no longer occur.
Communicability
Viral shedding persists for 1 to 3 weeks. Infected persons are most likely to transmit HAV 1 to 2 weeks before the onset of illness, when HAV concentration in stool is highest. The risk then decreases and is minimal the week after the onset of jaundice.
Risk Factors
Groups at increased risk for hepatitis A or its complications include international travelers (particularly high-risk itineraries like travel to rural areas in high-risk countries), contacts of recent international adoptees from HAV endemic countries, men who have sex with men, and users of illegal drugs. Outbreaks of hepatitis A have also been reported among persons working with hepatitis A-infected primates. This is the only occupational group known to be at increased risk for hepatitis A.
Persons with chronic liver disease are not at increased risk of infection but are at increased risk of acquiring fulminant hepatitis A. Persons with clotting factor disorders may be at increased risk of HAV because of administration of solvent/detergent-treated factor VIII and IX concentrates.
Foodhandlers are not at increased risk for hepatitis A because of their occupation, but are noteworthy because of their critical role in common-source foodborne HAV transmission. Health-care personnel do not have an increased prevalence of HAV infections, and nosocomial HAV transmission is rare. Nonetheless, outbreaks have been observed in neonatal intensive care units and in association with adult fecal incontinence. Institutions for persons with developmental disabilities previously were sites of high HAV endemicity. However, as fewer children have been institutionalized, conditions within these institutions have improved, and more children have been vaccinated. HAV incidence and prevalence have decreased, but sporadic outbreaks can occur. Schools are not common sites for HAV transmission. Multiple cases among children at a school require investigation of a common source and efforts to improve vaccination coverage. No worker related HAV infection have been reported in the United States. Consistently, serologic studies in the US have shown no or mildly increased risk of HAV infection in wastewater workers.
Children play an important role in HAV transmission. Children generally have asymptomatic or unrecognized illnesses, so they may serve as a source of infection, particularly for household or other close contacts.
In 2010, 75% of hepatitis A cases (who responded to any question about risk behaviors and exposures) indicated no risk factors for their infection. Of cases indicating at least one risk factor 2-6 weeks prior to the onset of illness, the most frequently reported source of infection was personal contact (sexual or household) with an infected person (7.3%). Employment or attendance at a nursery, day-care center, or preschool involved 3.1% of cases; 4% involved contact with a child or employee in child care; 14.1% occurred among persons reporting recent international travel; and 10.4% occurred in the context of a recognized foodborne or waterborne outbreak. Injection-drug use was a reported risk factor in 2% of cases; men who have sex with men represented 4.9% of cases.
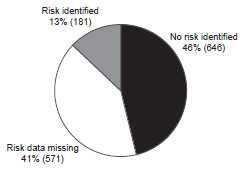
Of the 1,398 case reports of acute hepatitis A received by CDC during 2011, a total of 571 (41%) cases did not include a response (i.e. a “yes” or “no” response to any of the questions about risk behaviors and exposures) to enable assessment of risk behaviors or exposures. Of the 827 case reports that had a response, 646 (78%) indicated no risk behaviors/exposures for acute hepatitis A, and 181 (22%) indicated at least one risk behavior/exposure for acute hepatitis A during the 2-6 weeks prior to onset of illness.
Hepatitis A — United States, 2011
Hepatitis A — United States, 1966-2011
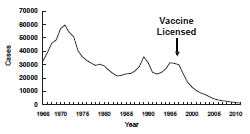
Secular Trends in the United States
Hepatitis A became nationally reportable as a distinct entity in 1966. During the prevaccine era in the United States, hepatitis A occurred in large nationwide epidemics.The largest number of cases reported in one year was in 1971 (59,606) and the last increase in cases occurred from 1994 to 1995. Prior to 2000, the incidence of reported hepatitis A was substantially higher in the western United States than in other parts of the country. From 1987 to 1997, 11 mostly western states (Arizona, Alaska, Oregon, New Mexico, Utah, Washington, Oklahoma, South Dakota, Idaho, Nevada, California) accounted for 50% of all reported cases but only 22% of the U.S. population. Historically, children 2 through 18 years of age have had the highest rates of hepatitis A (15 to 20 cases per 100,000 population in the early to mid-1990s).
In 1996, CDC’s Advisory Committee on Immunization Practices (ACIP) recommended administration of hepatitis A vaccine to persons at increased risk for the disease, including international travelers, men who have sex with men, non-injection and injection-drug users, and children living in communities with high rates of disease. In 1999, ACIP also recommended routine vaccination for children living in 11 Western states with average hepatitis A rates of >20 cases per 100,000 population and recommended that vaccination be considered for children in an additional six states with rates of 10-20 cases per 100,000 population. ACIP expanded these recommendations in 2006 to include routine vaccination of children in all 50 states.
Hepatitis A rates have been declining since vaccination initiation in 1996, and since 1998 have been at historically low levels.The number of reported acute hepatitis A cases decreased 93.7% overall from 1990 to 2009, and the last increase in cases occurred from 1994 to 1995. Many of the high-incidence states began routine hepatitis A vaccination programs for children in the late 1990s and since 2002, rates have been similar in all parts of the country, ranging from 0.1 case per 100,000 population in Arkansas, Mississippi, and South Dakota to 1.0 case per 100,000 population in Arizona. Since 2002, rates among children have declined and are now similar to other age groups. The wider use of vaccine is largely responsible for the marked decrease in hepatitis A rates in the United States and similar rates of infection throughout the country, and decreased infection rates in children. Beginning in the late 1990s, national age-specific rates declined more rapidly among children than adults; as a result, in recent years, rates have been similar among all age groups. Historic differences in rates among racial/ethnic populations also have narrowed in the vaccine era.
In 2010, a total of 1,670 cases of acute hepatitis A were reported nationwide to CDC. The overall incidence rate for 2010 was 0.5 cases per 100,000 population. The rate was similar among all age groups and gender. However, beginning in 2008, rates among Asian Pacific Islanders were higher than those among all other racial/ethnic populations. Based on data from the National Health and Nutrition Examination Survey (NHANES) conducted from 1999 through 2006, the overall seroprevalence of total antibody to HAV (anti-HAV) among the general U.S. population was 34.9% and 28.1% among U.S.-born individuals alone. Seroprevalence of HAV antibody increases with age, from 22.9% among 6- to 11-year-olds to 59.7% among persons 60 years of age and older. In this survey, anti-HAV seroprevalence was highest among Mexican Americans not born in the U.S. regardless of age, and seroprevalence was higher among U.S.-born Mexican Americans compared with U.S-born non-Hispanic white and non-Hispanic black persons for all age groups. Asian Pacific Islanders were not included as a race/ethnic category in this survey. The 1988 to 1994 NHANES total population age-adjusted seroprevalence of anti-HAV was not significantly different from the 1999-2006 age-adjusted seroprevalence. However, the overall age-adjusted seroprevalence increased among U.S. born children (6-19 years) during 1999-2006 compared to 1988-2004 from 8% to 20.2%. In addition, for individuals younger than 40 years, seroprevalence was higher in vaccinating states compared to non-vaccinating states for all age groups. This suggests increased hepatitis A vaccination rates following the 1999 ACIP recommendations.
The rate of hospitalization for hepatitis A in the United States declined more than 68% from the pre- to post-vaccine era (1996-2004) for all age groups. Similarly the rate of ambulatory care visits declined more than 40%. Medical expenditures for both hospitalizations and ambulatory care visits were estimated to have declined by approximately 68% ($29.1 to $9.3 million).
Case Definition
The 2012 case definition for hepatitis A was approved by the Council of State and Territorial Epidemiologists (CSTE) and published in a 2011 position statement. The clinical description for acute hepatitis A is an acute illness with a discrete onset of any sign or symptom consistent with acute viral hepatitis (e.g., fever, headache, malaise, anorexia, nausea, vomiting, diarrhea, and abdominal pain), and either a) jaundice, or b) elevated serum alanine aminotransferase (ALT) or aspartate aminotransferase (AST) levels. Since HAV cannot be differentiated from other types of viral hepatitis on clinical or epidemiologic features alone, serologic evidence of HAV-specific antibody is necessary. The diagnosis of acute hepatitis A requires the presence of HAV-specific IgM antibody.
Hepatitis A Vaccine
Hepatitis A Vaccines
- Inactivated whole-virus vaccines
- Pediatric and adult formulations
- pediatric formulations approved for persons 12 months through 18 years
- adult formulations approved for persons 19 years and older
Characteristics
Two inactivated whole-virus hepatitis A vaccines are available: HAVRIX (GlaxoSmithKline) and VAQTA (Merck). To produce each vaccine, cell culture-adapted virus is propagated in human fibroblasts, purified from cell lysates, inactivated with formalin, and adsorbed to an aluminum hydroxide adjuvant. HAVRIX is prepared with a preservative (2-phenoxyethanol); VAQTA does not contain a preservative. HAVRIX is available in two formulations: pediatric (720 ELISA units [EL.U.] per 0.5-mL dose) and adult (1,440 EL.U. per 1.0-mL dose). VAQTA is also available in two formulations: pediatric 0.5ml (25U of antigen) and adult 1.0ml (50U of antigen) formulations. The pediatric formulations of both vaccines are approved for persons 12 months through 18 years. The adult formulations are approved for persons 19 years and older. Both vaccines are approved for 2-dose schedules. The second dose of VAQTA is administered 6-18 months after the first dose, and the second dose of HAVRIX is administered 6-12 months after the first dose.
Hepatitis A Vaccine Immunogenicity
- Adults
- more than 95% seropositive after one dose
- nearly 100% seropositive after two doses
- Children and Adolescents
- more than 97% seropositive after one
- 100% seropositive after 2 doses (in clinical trials)
Hepatitis A Vaccine Efficacy
- HAVRIX
- 40,000 Thai children 1to 16 years of age
- vaccine efficacy 94%
- VAQTA
- 1,000 New York children 2 to 16 years of age
- vaccine efficacy 100%
Immunogenicity and Vaccine Efficacy
Both monovalent hepatitis A vaccines are highly immunogenic. More than 95% of adults will develop protective antibody within 4 weeks of a single dose of either vaccine, and nearly 100% will seroconvert after receiving two doses. Among children and adolescents, more than 97% will be seropositive within a month of the first dose. In clinical trials, all recipients had protective levels of antibody after two doses.
Both vaccines are effective in preventing clinical hepatitis A. The efficacy of HAVRIX in protecting against clinical hepatitis A was 94% among 40,000 Thai children 1 to 16 years of age who received two doses 1 month apart while living in villages with high HAV disease rates. The efficacy of VAQTA in protecting against clinical hepatitis A was 100% among 1,000 New York children 2 to 16 years of age who received one dose while living in a community with a high HAV disease rate.
Ten year follow-up data of serial anti-HAV levels after two doses of inactivated hepatitis A vaccine is available. A study in Alaska Native/American Indian individuals has shown that seropositivity for hepatitis A persists for at least 10 years after completing the two-dose vaccination at age 12 to 21 months, regardless of maternal anti-HAV status. Data from two other studies using the same population showed that protective anti-HAV levels persist 15 and 17 years after receiving three doses of a lower antigen content, inactivated hepatitis A vaccine starting at ages 3-6 years. Sustained protection will continue to be assessed by persistence of anti-HAV.
Vaccination Schedule and Use
ACIP Recommendation for Routine Hepatitis A Vaccination of Children
- All children should receive hepatitis A vaccine at 12 through 23 months of age
- Vaccination should be integrated into the routine childhood vaccination schedule
- Children who are not vaccinated by 2 years of age can be vaccinated at subsequent visits
- States, counties, and communities with existing hepatitis A vaccination programs for children 2 through 18 years of age should maintain these programs
- New efforts focused on routine vaccination of children 12 months of age should enhance, not replace ongoing vaccination programs for older children
- In areas with without an existing hepatitis A vaccination program catch-up vaccination of unvaccinated children 2 through 18 years of age can be considered
Following its introduction in 1996, hepatitis A vaccine was initially recommended for children and adolescents in communities with high or intermediate HAV endemicity. While this strategy prevented infection in high risk areas of the United States, it had little or no impact on the incidence of HAV infection in the United States.
All children should receive hepatitis A vaccine at age 1 year (i.e., 12 through 23 months). Vaccination should be completed according to the licensed schedules and integrated into the routine childhood vaccination schedule. Children who are not vaccinated by age 2 years can be vaccinated at subsequent visits. States, counties, and communities with existing hepatitis A vaccination programs for children aged 2 through18 years are encouraged to maintain these programs. In these areas, efforts should focus on routine vaccination of children 12 months of age and should enhance, not replace, ongoing programs directed at a broader population of children. In areas without existing hepatitis A vaccination programs, catch-up vaccination of unvaccinated children aged 2 through 18 years can be considered. Such programs might especially be warranted in the context of increasing incidence or ongoing outbreaks among children or adolescents.
Adults 19 years of age and older receive the adult formulation of hepatitis A vaccine according to licensed schedules. Persons at increased risk for HAV infection, or who are at increased risk for complications of HAV infection, should be routinely vaccinated.
For children less than 2 years of age, the vaccine should be administered intramuscularly into the anterolateral area of the thigh. For adults, the vaccine should be administered intramuscularly into the deltoid muscle. A needle length appropriate for the person’s age and size (minimum of 1 inch) should be used.
Limited data indicate that vaccines from different manufacturers are interchangeable. Completion of the series with the same product is preferable. However, if the originally used product is not available or not known, vaccination with either product is acceptable.
For both vaccines, the dosage of the 2nd dose should be based on the person’s age at the time of the dose, not the age when the first dose was given. For example, if a person received the first dose of the pediatric formulation of VAQTA at 18 years of age, and returns for the second dose at age 19 years, the second dose should be the adult formulation, not the pediatric formulation.
Hepatitis A Vaccines
| Formulation | HAVRIX | VAQTA |
|---|---|---|
| Pediatric | ||
| Age | 1 through 18 years | 1 through 18 years |
| Volume | 0.5 mL | 0.5 mL |
| Dose | 720 (EL.U) | 25 U |
| Schedule* | 0, 6-12 | 0, 6-18 |
| Number of Doses | 2 | 2 |
| Adult | ||
| Age | 19 years and older | 19 years and older |
| Volume | 1.0 mL | 1.0 mL |
| Dose | 1,440 (EL.U) | 50 U |
| Schedule* | 0, 6-12 | 0, 6-18 |
| Number of Doses | 2 | 2 |
The minimum interval between the first and second doses of hepatitis A vaccine is 6 calendar months. If the interval between the first and second doses of hepatitis A vaccine extends beyond 18 months, it is not necessary to repeat a dose.
Combination Hepatitis A and Hepatitis B Vaccine
Twinrix
- Combination hepatitis A vaccine (pediatric dose) and hepatitis B (adult dose)
- Schedules
- 0, 1, 6 months, or
- 0, 7, 21to 30 days and a booster dose 12 months after first dose
- Approved for persons 18 years of age and older
Persons at Increased Risk for Hepatitis A or Severe Outcomes of Infection
- International travelers
- Close contact with an international adoptee from a country of high or intermediate endemicity
- Men who have sex with men
- Persons who use illegal drugs
- Persons who have a clotting factor disorder
- Persons with occupational risk
- Persons with chronic liver disease
- Healthcare workers: not routinely recommended
- Child care centers: not routinely recommended
- Sewer workers or plumbers: not routinely recommended
- Food handlers: may be considered based on local epidemiology
In 2001, the Food and Drug Administration (FDA) approved a combination hepatitis A and hepatitis B vaccine (Twinrix, GlaxoSmithKline). Each dose of Twinrix contains 720 EL.U. of hepatitis A vaccine (equivalent to a pediatric dose of HAVRIX), and 20 mcg of hepatitis B surface antigen protein (equivalent to an adult dose of Engerix-B). The vaccine is administered in a three-dose series at 0, 1, and 6 months. Appropriate spacing of the doses must be maintained to assure long-term protection from both vaccines. The first and second doses should be separated by at least 4 weeks, and the second and third doses should be separated by at least 5 months. Twinrix is approved for persons aged 18 years and older and can be used in persons in this age group with indications for both hepatitis A and hepatitis B vaccines.
In 2007, FDA approved an alternative schedule for Twinrix with doses at 0, 7, and 21 through 30 days and a booster dose 12 months after the first dose.
Because the hepatitis B component of Twinrix is equivalent to a standard dose of hepatitis B vaccine, the schedule is the same whether Twinrix or single-antigen hepatitis B vaccine is used.
Single-antigen hepatitis A vaccine may be used to complete a series begun with Twinrix and vice versa. A person 19 years of age or older who receives one dose of Twinrix may complete the hepatitis A series with two doses of adult formulation hepatitis A vaccine separated by at least 5 months. A person who receives two doses of Twinrix may complete the hepatitis A series with one dose of adult formulation hepatitis A vaccine or Twinrix 5 months after the second dose. A person who begins the hepatitis A series with single-antigen hepatitis A vaccine may complete the series with two doses of Twinrix or one dose of adult formulation hepatitis A vaccine. An 18-year-old should follow the same schedule using the pediatric formulation.
Persons at Increased Risk for Hepatitis A or Severe Outcomes of Infection
Persons at increased risk for hepatitis A should be identified and vaccinated. Hepatitis A vaccine should be strongly considered for persons 1 year of age and older traveling to or working in countries where they would have a high or intermediate risk of hepatitis A virus infection. These areas include all areas of the world except Canada, Western Europe and Scandinavia, Japan, New Zealand, and Australia.
The first dose of hepatitis A vaccine should be administered as soon as travel is considered. For healthy persons 40 years of age or younger, 1 dose of single antigen vaccine administered at any time before departure can provide adequate protection.
Unvaccinated adults older than 40 years of age, immunocompromised persons, and persons with chronic liver disease planning to travel in 2 weeks or sooner should receive the first dose of vaccine and also can receive immune globulin at the same visit. Vaccine and IG should be administered with separate syringes at different anatomic sites.
Travelers who choose not to receive vaccine should receive a single dose of IG (0.02 mL/kg), which provides protection against HAV infection for up to 3 months. Persons whose travel period is more than 2 months should be administered IG at 0.06 mL/kg. IG should be repeated in 5 months for prolonged travel.
In 2009 ACIP recommended hepatitis A vaccination for all previously unvaccinated persons who anticipate close personal contact (e.g., household contact or regular babysitting) with an international adoptee from a country of high or intermediate endemicity during the first 60 days following arrival of the adoptee in the United States. The first dose of the 2-dose hepatitis A vaccine series should be administered as soon as adoption is planned, ideally 2 or more weeks before the arrival of the adoptee.
Other groups that should be offered vaccine include men who have sex with other men, persons who use illegal drugs, persons who have clotting factor disorders, and persons with occupational risk of infection. Persons with occupational risk include only those who work with hepatitis A-infected primates or with hepatitis A virus in a laboratory setting. No other groups have been shown to be at increased risk of hepatitis A infection due to occupational exposure.
Persons with chronic liver disease are not at increased risk for HAV infection because of their liver disease alone. However, these persons are at increased risk for fulminant hepatitis A should they become infected. Susceptible persons who have chronic liver disease should be vaccinated. Susceptible persons who either are awaiting or have received liver transplants should be vaccinated.
Hepatitis A vaccination is not routinely recommended for healthcare personnel, persons attending or working in child care centers, or persons who work in liquid or solid waste management (e.g., sewer workers or plumbers). These groups have not been shown to be at increased risk for hepatitis A infection. ACIP does not recommend routine hepatitis A vaccination for food service workers, but vaccination may be considered based on local epidemiology.
Prevaccination Serologic Testing
Hepatitis A Serologic Testing
- Prevaccination
- not indicated for children
- may be considered for some adults and older adolescent
- Postvaccination
- not indicated
HAV infection produces lifelong immunity to hepatitis A, so there is no benefit of vaccinating someone with serologic evidence of past HAV infection. The risk for adverse events following vaccination of such persons is not higher than the risk for serologically negative persons. As a result, the decision to conduct prevaccination testing should be based chiefly on the prevalence of immunity, the cost of testing and vaccinating (including office visit costs), and the likelihood that testing will interfere with initiating vaccination.
Testing of children is not indicated because of their expected low prevalence of infection. Persons for whom prevaccination serologic testing will likely be most cost-effective include adults who were either born in or lived for extensive periods in geographic areas that have a high endemicity of HAV infection (e.g., Central and South America, Africa, Asia); older adolescents and adults in certain populations (i.e., American Indian/Alaska Native and Hispanic); adults in certain groups that have a high prevalence of infection, and adults 40 years of age and older.
Commercially available tests for total anti-HAV should be used for prevaccination testing.
Postvaccination Serologic Testing
Postvaccination testing is not indicated because of the high rate of vaccine response among adults and children. Testing methods sufficiently sensitive to detect low anti-HAV concentrations after vaccination are not approved for routine diagnostic use in the United States.
Contraindications and Precautions to Vaccination
Hepatitis A Vaccine Contraindications and Precautions
- Severe allergic reaction to a vaccine component or following a prior dose
- Moderate or severe acute illness
Hepatitis A vaccine should not be administered to persons with a history of a severe allergic reaction (e.g. anaphylaxis) to a vaccine component or following a prior dose of hepatitis A vaccine, hypersensitivity to alum or, in the case of HAVRIX, to the preservative 2-phenoxyethanol. Vaccination of persons with moderate or severe acute illnesses should be deferred until the person’s condition has improved.
The safety of hepatitis A vaccination during pregnancy has not been determined. However, because it is an inactivated vaccine, the theoretical risk to the fetus is low. The risk associated with vaccination should be weighed against the risk for HAV infection. Because hepatitis A vaccine is inactivated, no special precautions are needed when vaccinating immunocompromised persons, although response to the vaccine may be suboptimal.
Adverse Reactions Following Vaccination
Hepatitis A Vaccine Adverse Reactions
- Local reaction
- 20%-50%
- Systemic reactions (malaise, fatigue)
- <10%
- No serious adverse reactions reported
For both vaccines, the most commonly reported adverse reaction following vaccination is a local reaction at the site of injection. Injection site pain, erythema, or swelling is reported by 20% to 50% of recipients. These symptoms are generally mild and self-limited. Mild systemic complaints (e.g., malaise, fatigue, low-grade fever) are reported by fewer than 10% of recipients. No serious adverse reactions have been reported.
Vaccine Storage and Handling
Hepatitis A vaccine should be maintained at refrigerator temperature between 35°F and 46°F (2°C and 8°C). Manufacturer package inserts contain additional information. For complete information on best practices and recommendations please refer to CDC’s Vaccine Storage and Handling Toolkit [4.33 MB, 109 pages].
Postexposure Prophylaxis
Immune globulin (IG) is typically used for postexposure prophylaxis of hepatitis A in susceptible persons. Hepatitis A vaccine may be used for postexposure prophylaxis in healthy persons 12 months through 40 years of age. Immune globulin is preferred for persons older than 40 years of age, children younger than 12 months of age, immunocompromised persons, and persons with chronic liver disease.
See MMWR 2007;56(No.41):1080-84 (October 19, 2007) for details.
Acknowledgement
The editors thank Drs. Trudy Murphy, Noelle Nelson, and Clive Brown, CDC for their assistance in updating this chapter.
Selected References
- Byrd KK, Bruden DL, Bruce MG, Bulkow LR, Zanis CL, Snowball MM, et al. Long term immunogenicity of inactivated hepatitis A vaccine: Follow-up at 15 years. Pediatric Infectious Disease Journal 2010;5:321-6.
- CDC. Update: prevention of hepatitis A virus after exposure to hepatitis A virus in international travelers. Updated recommendations of the Advisory Committee on Immunization Practices (ACIP). MMWR 2007;56:1080-84.
- CDC. Prevention of hepatitis A through active or passive immunization: recommendations of the Advisory Committee on Immunization Practices (ACIP). MMWR 2006;55(No. RR-7):1-23.
- CDC. Updated Recommendations from the Advisory Committee on Immunization Practices (ACIP) for Use of Hepatitis A Vaccine in Close Contacts of Newly Arriving International Adoptees. MMWR 2009;58;1006-7.
- CDC. Viral Hepatitis Surveillance United States 2010 [1.53 MB, 67 pages]. Accessed March 19, 2013.
- CDC. Hepatitis A outbreak associated with green onions at a restaurant—Monaca, Pennsylvania, 2003. MMWR 2003;52:1155-7.
- Council of State and Territorial Epidemiologists Position Statement, Infectious Disease, Public Health Reporting and National Notification for Hepatitis A [8 pages], 11-ID-02.
- Klevens RM, Kruszon-Moran D, Wasley A, Gallagher K, McQuillan GM, Kuhnert W, et al. Seroprevalence of hepatitis A virus antibodies in the U.S.: results from the National Health and Nutrition Examination Survey. Public Health Rep. 2011 Jul-Aug;126(4):522-32.
- Murphy TV, Feinstone SM, and Bell BP. Hepatitis A Vaccine. In: Plotkin SA, Orenstein, WA, and Offit PA, eds. Vaccines. 6th ed. China: Saunders; 2013.
- Raczniak GA, Bulkow LR, Bruce MG, Zanis CL, Baum RL, Snowball MM, et al. Long-term immunogenicity of hepatitis A virus vaccine in Alaska 17 years after initial childhood series. The Journal of infectious diseases, 2013, Vol.207(3), pp.493-6
- Sharapov UM, Bulkow LR, Negus SE, Spradling PR, Homan C, Drobeniuc J, et al. Persistence of hepatitis A vaccine induced seropositivity in infants and young children by maternal antibody status: 10-year follow-up. Hepatology. 2012 Aug;56(2):516-22. doi: 10.1002/hep.25687. Epub 2012 Jun 11.
- Taylor RM, Davern T, Munoz S, Han SH, McGuire B, Larson AM, et al. Fulminant hepatitis A virus infection in the United States: Incidence, prognosis, and outcomes. Hepatology. 2006 Dec;44(6):1589-97.
- Zhou F, Shefer A, Weinbaum C, McCauley M, Kong Y. Impact of hepatitis A vaccination on health care utilization in the United States, 1996-2004. Vaccine. 2007 May 4;25(18):3581-7. Epub 2007 Jan 25.
- Page last reviewed: November 15, 2016
- Page last updated: May 15, 2015
- Content source:


 ShareCompartir
ShareCompartir